
Shinichi Ozawa
AJINOMOTO Co., Inc.
Institute for Innovation

Hiroshi Miyano
AJINOMOTO Co., Inc.
Institute for Innovation
Coauthor:
Masahito Ito*1
Approximately 60% of the human body is water, but half of the rest, approximately 20% of the total, consists of proteins and other materials composed of amino acids. Thus, in the case of someone weighing 50 kg, approximately 10 kg is protein, and various essential tissues, including muscles, the gastrointestinal tract, and internal organs, are largely composed of protein, in addition to which the protein hemoglobin is an important constituent of blood, and collagen and other proteins are important constituents of hair and skin1). Even among the three primary macronutrient groups, in the human body, most of the fundamental nutrients that are essential for life activities are proteins. There are 20 amino acids that combine to form proteins, but numerous other amino acids are present in the human body, and they perform important roles in various biological activities, including metabolism, immunity, digestion, central nervous system function, growth and development, biological protection, and locomotion. Furthermore, taking the biosphere as a whole, a total of approximately 500 types of amino acid have been shown to exist.
In the present article, the history and current state of amino acid analysis are elucidated, focusing primarily on the L-8900 Amino Acid Analyzer, which, among the separation and analysis technologies that are indispensable analysis techniques in amino acid research, can be seen to be by far the best high-performance liquid chromatography (HPLC) device. In addition, the requirements for future analyzers are detailed.
Amino acid analysis has for a long time been carried out by microbial quantification methods, using Lactobacillus. However, chromatography has gradually come to be the mainstream approach to separation, and to qualitative and quantitative analysis.
In 1941, the UK biochemists Martin and Synge developed partition chromatography for amino acid analysis3), followed in 1944 by paper chromatography4), and they succeeded in using these techniques to separate complex mixtures of amino acids and peptides. In 1955, Sanger, making use of partition and paper chromatography methods, succeeded in determining the entire amino acid sequence for insulin.
From 1948, the US biochemists Stein and Moore engaged in research on amino acid analysis by chromatography using starch particles and ion-exchange resins, and in 1958 they developed an automatic amino acid analyzer in collaboration with Spackman5). The basic principle of this analyzer has hardly been changed in the amino acid analyzers that are currently in widespread use (see below), and, as a general analysis method for the amino acids making up proteins, it has been found to be much better than the methods that were in previous use, in terms of sample volumes, analysis operations, etc. This analysis method was subsequently found to be of great value for determining the amino acid sequences in ribonuclease molecules.
Martin and Synge were awarded the Nobel Prize for their work in 1952, and Stein and Moore were in 19726, 7), showing that progress in amino acid analysis can be considered to have played a major role in the history of science.
The first amino acid analyzer in Japan was imported by Ajinomoto Co., Inc. (Ajinomoto). The world's first company to manufacture amino acid analyzers was Beckman Coulter, Inc., and in 1959 Ajinomoto purchased the second such device to be made. Incidentally, the first such device belonged to Merck and Co., and the third was installed at Rockefeller Research Laboratories, where the inventors, Moore and Stein, were working. Unfortunately, none of these devices still exist.
In 1963, Ajinomoto purchased a KLA-2 amino acid analyzer, which was being sold by Hitachi, Ltd.
According to published memoirs8), the analyzers at that time were dual-column devices, with a short column, for basic amino acid analysis, that had an internal diameter of 0.9 cm, and was 15 cm long; and a long column, for neutral and acidic amino acid analysis, that was 150 cm long. The buffer flow rate was 30 mL/hour, so basic amino acids were analyzed in the morning, and neutral and acidic amino acids were analyzed in the afternoon. In other words, a whole day was needed for analysis of the 17 types of amino acid found in protein hydrolysates.
Most amino acids do not have characteristic ultraviolet or visible-light absorption spectra, and are not fluorescent. Therefore, in general, the amino groups are selectively modified to form substances showing fluorescence or visible-light absorption, or derivatized, and then detected9).
Derivatization can be either by a pre-column derivatization method, in which the substances to be analyzed in the sample are derivatized before injection into the chromatography column, and the reaction components are then separated and detected; or by a post-column derivatization method, in which the substances to be analyzed in the sample are first separated in the column, followed by inline derivatization of the eluate, and then detection. In the below table, the characteristics of pre- and post-column derivatization methods are compared. Some reagents are solely for use with one of these methods, but there are others that can be applied to either10).
| Feasible with general HPLC devices. | Complex devices needed. |
|---|---|
| Special devices needed for automation. | Automation can be achieved easily. |
| Feasible even if a prolonged time is needed for the derivatization reaction, but in that case the analysis duration is prolonged. | The derivatization reaction must be reasonably rapid, and the overall analysis duration is reduced. |
| In setting reaction conditions, little consideration need be given to column elution conditions. | Elution conditions are markedly affected by the reaction conditions. |
| The reaction is quantitative. | Detection is possible even while the reaction is in progress. |
| There is a single reactive component. | Detection is possible even with multiple/complex reactive components. |
| The reaction component is relatively stable. | Detection is possible even with somewhat unstable reaction components. |
| It is acceptable if the reagent blank (excess reagent and/or separated reagent) is detected, as the target substance can be clearly separated. →The reagent is therefore selected on the basis of the detector type. |
It is essential to select a reagent blank that is not, or is only very slightly, detected by the detector. →The types of derivatization reagent that can be used are therefore restricted. |
| The amount of reagent consumed is small. | The amount of reagent consumed is large. |
| Dilution of the sample by the reagent solution can generally be disregarded. | Dilution of the sample by the reagent solution, and peak broadening, occur. |
| High sensitization is feasible. | Highly quantitative and reproducible. |
| The reaction efficiency of derivatization is affected by the sample matrix. | The sample components are separated in the column before the reaction. The sample matrix therefore has no effect at the time of reaction with the derivatized reagents. |
| Reverse-phase HPLC is applicable. | Reverse-phase HPLC is not applicable. |
Partly modified from Ref. 10 (Q55, Table 1).
The L-8900 Amino Acid Analyzer that is the central topic of this article has the same basic principle as the device developed by Stein and Moore. It involves a post-column derivatization method that uses ninhydrin as the analysis reagent, and it is used to detect the reactants of analysis-target substances formed after the reaction, and components that react with the reagent17)(Fig.1).
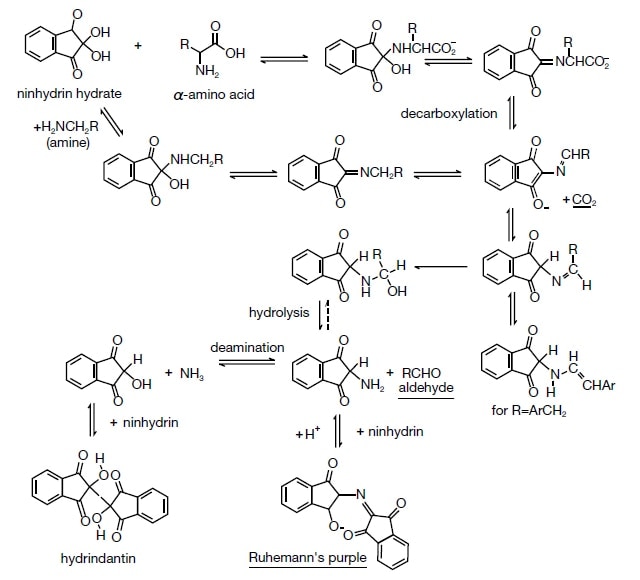
Fig. 1 Ninhydrin structure and colorization mechanism
In pre-column-derivatization amino acid analysis, it is generally the amino group of the amino acids that is derivatized.
Pre-treatment to make the derivative fluorescent is frequently carried out. The objectives of this include the following:
As shown in Fig. 2, various commercially available and well-known compounds are in use as derivatization reagents, including ortho-phthalaldehyde (OPA; λex.: 245 nm; λem.: 395 nm), 6-aminoquinolyl-N-hydroxysuccinyl imide (AQC; λex.: 245 nm; λem.: 395 nm), 4-fluoro-7-nitrobenzofurazane (NBD-F; λex.: 470 nm; λem.: 530 nm), 9-fluorenylmethyl chloroformate (FMOC; λex.: 340 or 350 nm; λem.: 450 nm), and fluorescein-4-cyanate (FITC; λex.: 390 nm; λem.: 520 nm). In addition, numerous other compounds have been developed, and a number of reviews of these have been published11, 12). However, some reagents, such as OPA, react only with primary amines, and care is therefore needed in this respect.
In connection with progress in liquid chromatography / mass spectrometry (LC/MS) and LC / tandem mass spectrometry (LC/MS/MS) in recent years, pre-column derivatization reagents have been developed for these techniques as applied to amino acid analysis.
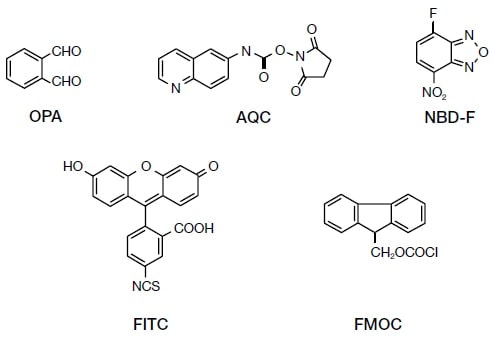
Fig. 2 Molecular structures of representative pre-column derivatization reagents
Fluorescent derivatization reagents used to require molecular structures such as to make the amino acid fluorescent, whereas in the case of many reagents for MS analysis, substructures that increase the ionization efficiency are provided (Fig. 3). In the case of MS/MS in particular, the signal/noise ratio is markedly increased. There have also been a number of reports about making full use of MS/MS, so as to achieve regular derivative cleavage, and thus to develop various analysis methods making use of these advances; and also about the feasibility of ultra-rapid measurement (for details, see references 13) to 15).
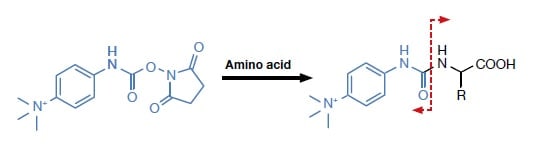
Fig. 3 Molecular structure of the pre-column derivatization reagent for LC/MS/MS (p-N,N,N-trimethylammonioanilyl N'-hydroxysuccinimidyl carbamate iodide; TAHS), the resulting derivative, and the cleavage sites in MS/MS
Since KLA-2 was launched by Hitachi in the 1960s (Fig. 4), various analyzers based on the post-column derivatization method, using ninhydrin as the reagent, have been developed. Since Model 835, the devices have been computer-controlled from sample injection to data-processing, and have had single stainless steel columns, and pumps that use ball valves.
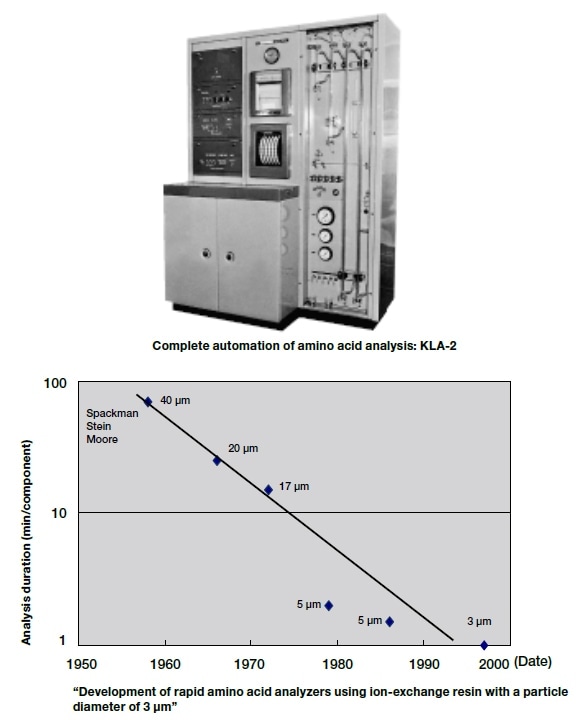
Fig. 4 History of Hitachi Amino Acid Analyzers
In 2005, Hitachi High-Tech Corporation (Hitachi High-Tech) and Ajinomoto collaboratively developed the High-speed Amino Acid Analyzer, Model L-8900, and launched it commercially. The L-8900 is a successor to the L-8800, and, although no changes were made with respect to precision or the number of amino acids that can be analyzed, proposals with respect to development concept and product specifications were made, from the point of view of user friendliness, mostly by Ajinomoto, and were then actualized by Hitachi High-Tech. In other words, the L-8800 was an analyzer with very high performance and general completeness, but from the point of view of the operator it was not easy to use, being a complicated and user-unfriendly device, so these disadvantages were resolved as far as possible in the L-8900.
In concrete terms, the analyzer in use previously had high performance levels, but there were disadvantages with its operation, including the length of time needed to achieve expertise, the high frequency of problems due to operational mistakes, and the length of time for which the operators had to be physically present. In this context, in 2004, Ajinomoto proposed various measures, including introduction of advanced touch panels and audio-guides; measures to enable analysis to be initiated by means of simple processes, on the basis of automation of the nitrogen valve, drainage valve, etc.; and introduction of mass sensors that prevent exhaustion of eluent. Hitachi High-Tech then put these proposals into practice as far as possible.
In addition, it was predicted that, as analyzers become easier to operate, the market demands for amino acid analysis would tend to increase, and the number of amino acids measured would increase, so, by using twin columns, a measurement method was developed in which the time per cycle needed for biological analysis was reduced from 150 to 75 minutes. In this twin-column device, two columns are connected in series, each being 5.4 mm in internal diameter, and 25 mm long. The pressure resistance of ion-exchange resin is limited, so, by using two columns, the flow rate can be increased to the limit of pressure resistance (actually the pressure differential) of each column. The outflow rate is 0.56 mL/min, and the column temperature can be set at any point between 28 and 70°C, using a time program.
The L-8900 High-speed Amino Acid Analyzer is a post-column-derivatization, high-speed LC device that uses columns packed with polystyrene divinylbenzene modified by sulfonate groups, this being a strongly acidic cation-exchange resin with particles 3 µm in diameter. The mobile phases used are sodium-citrate-type buffers for the 20 constituent amino acids, in the protein hydrolysate analysis method; and lithium-citrate-type buffer solution for the 40 constituent amino acids, in the biological fluid analysis method. Commercially available bottles of the buffer solutions can be attached to the device without modification. In either analysis method, in order to elute large numbers of components, a stepwise elution method is usually used, with various types of buffer solutions.
Each mobile phase is transferred by nitrogen gas, from a pressurized reagent bottle via an electromagnetic valve, and then by mobile phase pump 1. This pump is capable of transferring six liquids, including the regenerant. In order to suppress baseline noise in the chromatogram, pulsating flow, including pressure changes, is reduced using a one-stroke, 10 µL micropump. By synchronizing the pump-plunger suction process, and controlling the opening and closing times of each electromagnetic valve, each of the mobile phases can be mixed together in defined proportions.
The autosampler can be set so as to have up to 200 samples in 1.5 mL vials, and 20 µL aliquots of sample are taken up by suction, in a specified cycle, and injected into the separation column. If the sample volume is small, 200 µL microvials can be used. In addition, if the analysis type requires refrigerated storage, with inclusion of glutathione, etc., a 200 vial refrigerated rack is also included (Fig. 5).

Fig. 5 Photograph of L-8900
The sample is as detailed above, and is separated into amino acid components, with the separation mode being by means of a cation-exchange column (Fig. 6).
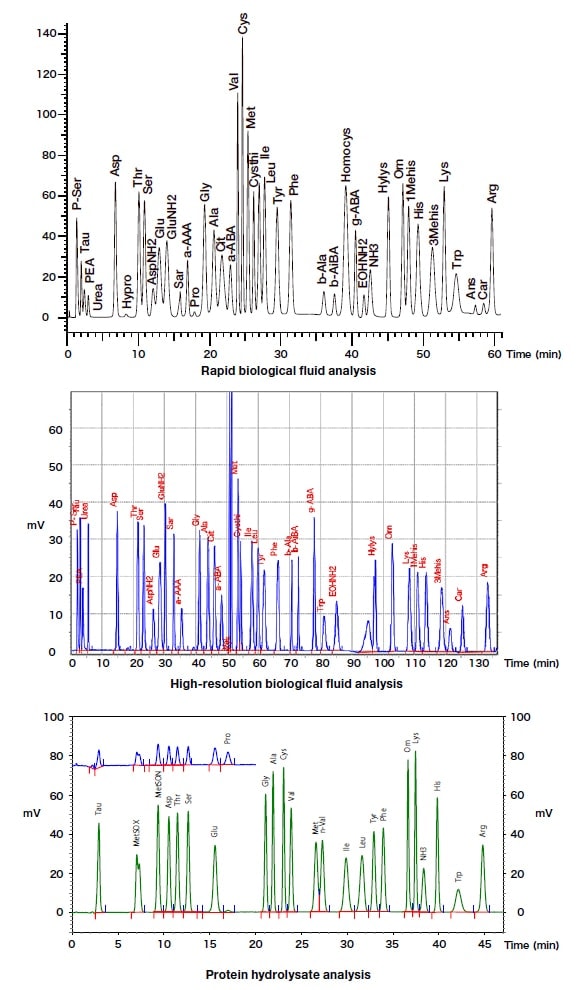
Fig. 6 Representative chromatogram obtained using L-8900
Packing materials for the separation column, and packed columns of different sizes, can be selected on the basis of the analysis method. In general, the outflow rate is 0.4 mL/min, and rapid, high-resolution analysis can be achieved with application of a pressure of approximately 10 MPa. A constant-column-temperature device maintains the separation column at constant temperature using a Peltier device. In addition, rapid heating and cooling control within the 20 to 85°C range can be achieved using a timer program.
For the ninhydrin reaction solution, two commercially available bottles containing reagents are attached to the device without modification, and the contents are mixed immediately before the reaction. Each of the amino acid components eluted from the column is mixed with reaction solution provided by reaction solution pump 2, and is heated to 135°C by the reaction device. In order to markedly suppress peak broadening while the mixture is flowing, a reaction column packed with inert beads is used as the reaction locus, instead of a fluorine-based resin tube.
The amino acids are detected at a wavelength of 570 nm, which is within the visible light spectrum, and is the absorbance maximum for Ruhemann's purple, which is one of the reaction components. Quantification is carried out on the basis of the chromatogram peaks. Proline and hydroxyproline, which are imino acids, have no absorbance maxima within the visible light spectrum, and are therefore detected at 440 nm. Visible light at a wavelength of 700 nm is used as the detection reference, and compensation for fluctuation of the halogen-tungsten lamp light source, etc., is made.
The ease of operation of L-8900 has been greatly increased, but it seems that we are only part of the way toward development of the ideally user-friendly device.
Ajinomoto has dozens of amino acid analyzers, both in Japan and overseas, and the devices are used by various operators, in diverse situations. Using the in-house network, we have independently developed navigation systems for use by these various operators, to enable them to access all essential information promptly. An example of such a system is as follows:
As the system is described above, one may imagine that it represents the systematization of Ajinomoto's know-how. However, in fact, in most cases it involves no more than full comprehension of data that are already available. Looking at this from the opposite perspective, many of the usable data and information items that users may want are already available if they are actually sought.
The "secrets" for doing this can be presented as follows:
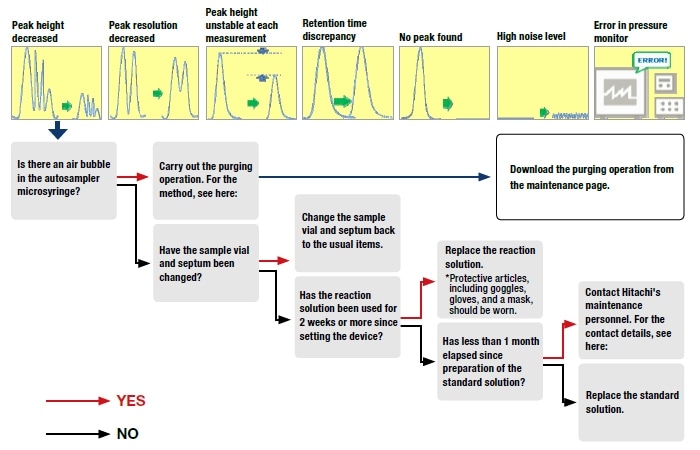
Fig. 7 Troubleshooting flowchart for "Peak height decreased", in the troubleshooting flow sheet in the in-house navigation system
The L-8900 Amino Acid Analyzer is now accepted as the reigning champion of post-column-derivatization HPLC devices, and successors are not expected to be superior with respect to precision and accuracy. However, in terms of user friendliness, the manufacturers have hopes for further improvement, as detailed below.
Users, including Ajinomoto have various purposes in using the device, control the end points of development, and have valuable ideas. Manufacturers have a great store of technological knowledge and information. It is important for users and manufacturers to pool their knowledge and opinions to aim high, and to achieve development of excellent devices. L-8900 is one outcome of such development, but the definition of "excellent" differs between users.
In 2011, a new research organization, the Japan Society of Amino Acid Analysis, was established16), and it holds a conference once per year. Thus, every year, almost 100 people gather together, and various presentations are made, by both manufacturers and users, about the amino acids, peptides, proteins, and amino-acid-related substances that are analyzed. This is an excellent venue for users and manufacturers to get together and discuss progress and requirements in detail. We hope that, by making good use of such venues, better devices, and thus in turn better outcomes, can be achieved.
References
Coauthor
See more