
Yuko Nishimoto
Professor
Department of Chemistry Faculty of Science
Kanagawa University
The field of cultural heritage science, which uses scientific methods to study archaeological and historical materials such as excavated samples, consists of three primary branches: (1) research related to production sites, aging, and ancient environments, (2) research on deterioration mechanisms and other material properties, and (3) research targeting preservation and restoration. In all cases, non-destructive analytical methods are prioritized, and the taking of material samples is kept to an absolute minimum. However, many archaeological and historical materials have non-uniform compositions, with elemental concentrations that vary from point to point; moreover, the majority of such materials are in an advanced state of decay. For these reasons, results obtained from the analysis of only one portion of a sample are often of questionable value in reflecting the properties of the sample as a whole. In cases where destructive analyses are allowable, it is thus important to perform as many measurements as possible to obtain a large amount of information. Here we report elemental mapping images of several archaeological specimens, including (a) silver and copper Kan’ei tsuho coins used as currency in the late Edo period of Japan, and (b) natural whetstones used to sharpen swords.
Among the various types of silver coins used as currency during Japan's late Edo period, we considered three: Tempo ichibu-gin silver coins, Ansei ichibu-gin silver coins, and Kaei isshugin silver coins. Although the nominal mass standards for these silver coins have been tabulated, values obtained by chemical analyses are not publicly available.1,2) We also expect that the metal composition of the coins is non-uniform. Table 1 lists the coins considered in this study.
| Silver coins | Quality1,2) | Year first produced |
|---|---|---|
| Tempo ichibu-gin silver coins | Highest-quality silver: 98.86% | 1837 |
| Ansei ichibu-gin silver coins | Same quality as nickel silver: 89.3% | 1859 |
| Kaei isshugin silver coins | High-quality Nanryo silver: 98.71% | 1853 |
To investigate variations in the composition in different regions of the samples, we first used a wavelength-dispersive fluorescence X-ray analyzer. After cleaning and drying two coins of each type, we made measurements of the entire sample surface (front and back) as well as—for the Ansei and Tempo ichibu-gin silver coins—regions of Ø10 mm near the top and bottom of the coins. For the Tempo ichibu-gin silver coins, measurements of both samples showed element concentrations of 96% Ag, 0.3% Cu, and 1% Si. The Cu concentration varied from region to region on both the front and back sides of the coins, with a maximum of 1% and a minimum of 0.2%. For the Ansei ichibu-gin silver coins, we observed discrepancies between our two samples, with concentrations of 92–96% Ag, 3–5% Cu, and 0.2–2% Si. The Cu concentration varied from region to region with maximum and minimum values of 6.5% and 2.1%, respectively. For the Kaei isshugin silver coins, only measurements of the entire surface of the sample were performed, and these yielded results that differed between samples, with concentrations of 95–97% Ag, 0.5–0.7% Cu, and 0.5–1.5% Si.
We next restricted the range of measurements to regions of Ø2.5 mm and used an energy-dispersive X-ray fluorescence analyzer (Hitachi High-Tech Science, SEA5200A) to perform mapping measurements. The concentrations of Cu, Ag, Si, Sn, Pb, and As were found to vary depending on the region sampled. In particular, the concentrations of Ag and Si were correlated, with Ag-poor regions being Si-rich. Figures 1–3 show representative results of mapping measurements for the various samples.
To obtain further information, we shaved 0.2 g samples from the interior of one of the Ansei ichibu-gin silver coins and from the interior of one of the Tempo ichibu-gin silver coins, dissolved these samples by heating in sulfuric acid/ nitric acid, and then added dihydroxysuccinic acid to form a sample solution. Similarly, we shaved 0.2 g from one of the Kaei isshugin silver coins, dissolved the sample in nitric acid, and then dissolved any undissolved remainder with nitrohydrochloric acid. We carried out quantitative composition analysis on each sample using an inductively coupled plasma atomic emission spectrometer (ICP-AES) (Hitachi High-Tech Science, SPS3500). The Ag content of these samples, as determined by X-ray fluorescence analysis of the entire sample surface, was as follows: for the Tempo ichibu-gin silver coins: 95.9% (front) / 97.7% (back); for the Ansei ichibu-gin silver coins: 93.8% (front) / 91.4% (back); for the Kaei isshugin silver coins: 96.4% (front) / 97.1% (back). Discrepancies were observed in the concentrations of the trace elements Cu, Sb, Si, As, and Zn. All of these elements were present in significant quantities in the Ansei ichibu-gin silver coins. The results of the measurements are summarized in Table 2 as solid concentrations.
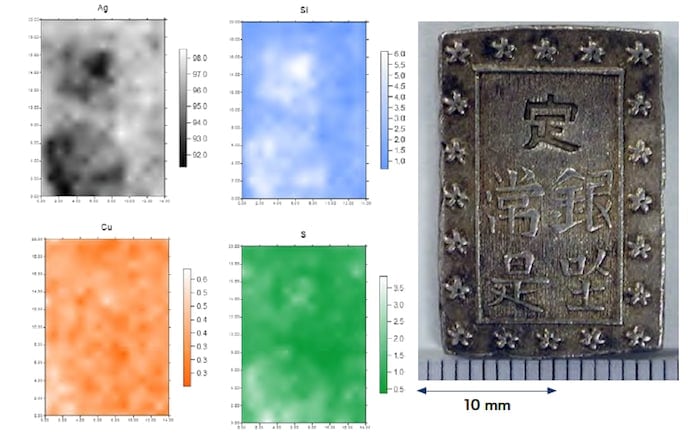
Fig.1 EDS mapping measurements of the back side of a Tempo ichibu-gin silver coin (Ø2.5 mm)
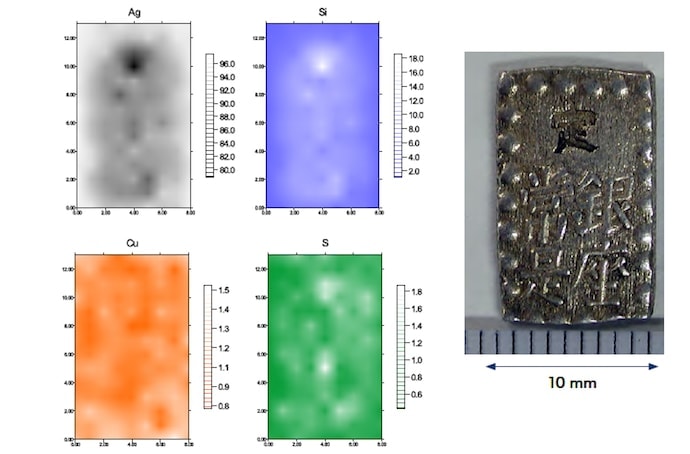
Fig.2 EDS mapping measurements of the back side of an Ansei ichibu-gin silver coin (Ø2.5 mm)
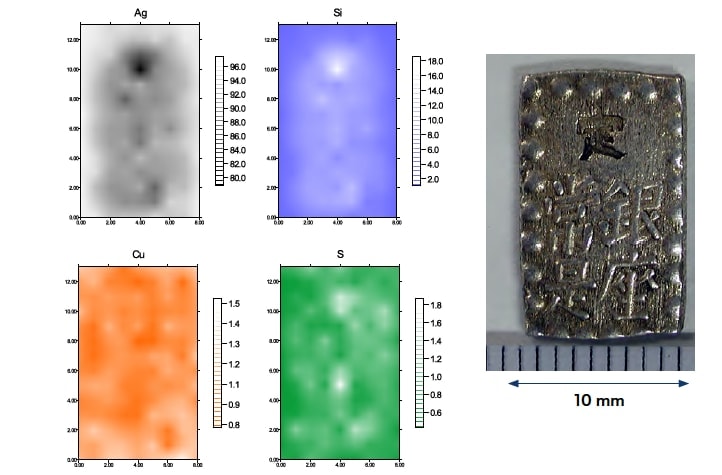
Fig.3 EDS mapping measurements of the back side of a Kaei isshugin silver coin (Ø2.5 mm)
| Element (lower bound) | Ansei ichibu-gin silver coin(10.1% total non-silver content) | Tempo ichibu-gin silver coin (0.4% total non-silver content) | Kaei isshugin silver coin (1.4% total non-silver content) |
|---|---|---|---|
| Cu (0.5) | 96192 | 1578 | 6895 |
| Pb (0.07) | 2072 | 1853 | 3211 |
| Zn (0.8) | 731 | 12 | 17 |
| Au (1.8) | 330 | 260 | 3017 |
| Sn (6.8) | 160 | 130 | 31 |
| Sb (10) | 430 | 20 | 2 |
| Al (2.4) | 38 | 40 | 20 |
| Ti (0.4) | 0.4 | <0.4 | 6.6 |
| As (18) | 180 | <18 | ND |
| Si (3) | 168 | 76 | 119 |
| Ca (1.3) | 89 | 101 | 133 |
| Mg (0.2) | <0.2 | <0.2 | 7.2 |
| Sr (0.02) | <0.02 | <0.02 | ND |
| Mn (0.07) | <0.07 | <0.07 | 0.3 |
| Na (0.6) | 110 | 116 | 311 |
| K (2.1) | 118 | 115 | 180 |
The results of all measurements consistently show that the Tempo ichibu-gin silver coins exhibit the greatest purity, while the Ansei ichibu-gin silver coins are the least pure. In the Ansei ichibu-gin silver samples, Cu—which constitutes some 10% of the coins—is plentiful in the interior, but is also scattered non-uniformly over the surface. When making measurements of coins, it is common to consider just one region of a sample; however, our findings demonstrate the importance of analyzing the entire sample combined with mapping measurements, particularly for the quantitative analysis of trace elements.
Copper coins made from a ternary alloy of Cu, Sn, and Pb were historically the most widely circulated form of currency. In the middle ages, the coins used as currency were brought in from overseas. Later, large numbers of duplicate and counterfeit coins were produced in regions throughout Japan, and these too went into circulation. For government-issued coins, it is thought that the relative concentrations of the raw materials—the base metals Cu, Sn, Pb—were adjusted over appropriate ranges to achieve certain standards for surface hardness, abrasion resistance, strength, ductility, and other material properties. The various metals were probably combined in the molten state to form a bronze alloy, which was then formed into coins. By contrast, in duplicate and counterfeit coins, the concentration of Sn is often extremely low.3-5) Whereas Cu and Pb mines were fairly numerous and widely distributed across mainland China and Japan, the geographical distribution of Sn was much sparser. It is thought that, before the modern era, Sn was produced in insufficient quantities and at higher manufacturing cost than other metals. The samples considered in this study are seven Kan’ei tsuho coins that were excavated in their entirety from Kokura Castle site 3: we denote these as #1 (old Kan’ei), #2 (new Kan’ei-genjisen), #3 (old Kan’ei), #4 (old Kan’ei), #5 (old Kan’ei), #6 (new Kan’ei), and #7 (new Kan’ei-monsen). It is believed that the circulation of Eiraku tsuho coins was terminated in 1608 (the 13th year of the Keicho era), and that the production of Kan’ei currency began in 1634 (the 11th year of the Kan’ei era). Near Kokura Castle stands Kurosaki Castle,6) which was constructed in the 9th year of the Keicho era and at which a counterfeit coin production facility was discovered.
| Sample number | Photograph of the coin | Sample number | Photograph of the coin |
|---|---|---|---|
| #1 (old Kan'ei) |
|
#5 (old Kan'ei) |
|
| #2 (new Kan'ei-genjisen) |
|
#6 (new Kan'ei) |
|
| #3 (old Kan'ei) |
|
#7 (new Kan'ei-monsen) |
|
| #4 (old Kan'ei) |
|
We first restricted the range of measurements to regions of Ø1 mm and used an energy-dispersive X-ray fluorescence analyzer to perform mapping measurements. Figures 4–6 show the results of measurements carried out for samples #4 and #6. For sample #4, the concentration distributions of Cu and Zn are similar, as are the distributions of As, Pb, and Fe. In addition, the distributions of the trace elements Co, Ti, and Si exhibit similar trends. For sample #6, the concentration distributions of Cu, Fe, and Zn are similar, as are the distributions of As, Pb, and S. The concentration distributions of As, Pb, and S are similar to those found for a counterfeit coin in which Sn was detected. These results illustrate the type of useful information that elemental analyses can provide. We next shaved off 25 mg samples, added 2 ml of hydrochloric acid and 0.5 ml of nitric acid, and subjected the samples to microwave heating inside a closed Teflon vessel. We then filtered the undissolved remainder with No. 5C filter paper and diluted the filtrate to a fixed volume of 50 ml or 100 ml, after which we performed quantitative composition measurements using ICP-AES. The results are listed in Table 4. For samples #1–4, the sum of all measured fractional compositions is less than 100%. For sample #3, we attribute this to the formation of a white condensate after dissolution. For sample #4, the primary culprit is the undissolved remainder after dissolution in acid (1.6 mg), but an additional factor is that sulfur—which is detected using X-ray fluorescence analysis—is not observed in these ICP-AES measurements.
All of the samples are copper coins for which the primary constituent is Cu. Samples #4 and #5 are a ternary alloy of Cu, Sn, and Pb. Samples #2 and #3 contain Fe and Pb as well as trace amounts of As and Sn. Sample #1 contains almost nothing except Cu and Pb.
| Sample | Cu | Pb | Fe | As | Sb | Sn | Ag | Al | Zn | Total |
|---|---|---|---|---|---|---|---|---|---|---|
| No.1 | 62.9 | 16.4 | 0.0 | <0 | 0.0 | 0.0 | 0.0 | <0 | 0.0 | 79.0 |
| No.2 | 63.8 | 6.6 | 3.8 | 1.7 | 0.3 | 0.7 | <0 | <0 | 0.0 | 76.9 |
| No.3 | 62.5 | 4.2 | 3.2 | 0.5 | 0.1 | 1.4 | 0.0 | <0 | 0.0 | 71.9 |
| No.4 | 40.5 | 12.4 | 0.9 | 0.0 | 0.0 | 7.8 | 0.1 | 0.8 | 0.0 | 97.7 |
| No.5 | 73.3 | 15.7 | 0.1 | 0.2 | 0.1 | 8.2 | 0.1 | <0 | 0.0 | 97.7 |
| No.6 | 81.1 | 17.8 | 1.2 | 0.9 | 0.0 | 0.0 | 0.0 | <0 | 0.1 | 101.1 |
| No.7 | 69.2 | 24.3 | 0.1 | 0.1 | 0.0 | 7.3 | 0.1 | <0 | 0.1 | 101.1 |
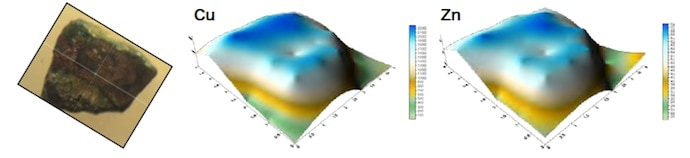
Fig.4 Results of measurements of copper coin sample #4 (Old Kan’ei)
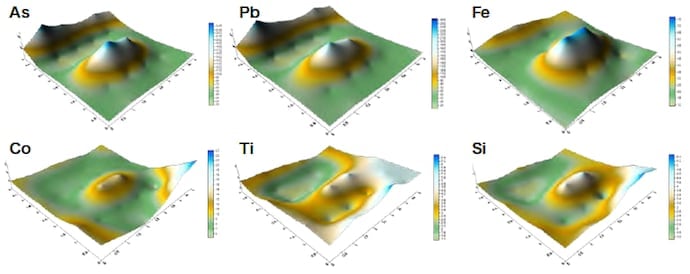
Fig.5 Results of measurements of copper coin sample #4
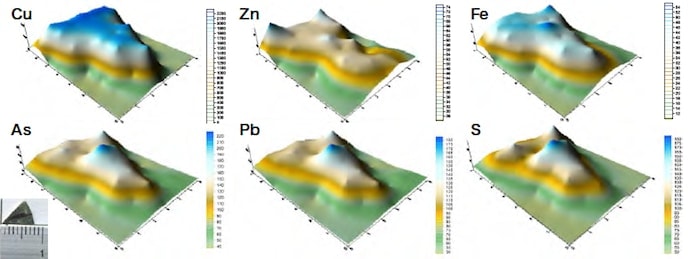
Fig.6 Results of measurements of copper coin sample #6
Natural whetstones are rocks suitable for sharpening purposes that are collected and resized appropriately to allow their surfaces to be used as sharpening planes. They are classified into the categories of rough, medium, and production-grade depending on their granularity. By combining the use of multiple whetstones of differing granularities it is possible to achieve efficient sharpening. We used fluorescence X-ray analysis to measure 26 whetstone samples used primarily to sharpen swords. Si was the most abundant element in all of the samples, with fractional concentrations ranging from 28 to 42%; the samples also contained Al, Fe, Ca, and K and trace quantities of Ti, Mn, Ba, Zr, Cr, V, and Cl. We estimated the reproducibility of our measurements to be within about 0.5% of the measured value for Si, 1% for Fe, and 0.8% for Ca. Based on a total of six measurements of five Aoto stones (one sample was measured in two regions of different colors), the concentrations of Fe, Si, and Ca were in the ranges of 3.4–4.8%, 32–37%, and 0.03–0.2%, respectively. Based on measurements of three Tsushima-do stones, (one sample was measured in two regions of different colors), the concentrations of Fe, Si, and Ca were found to be in the ranges of 4.3–5.1%, 28–30%, and 2.0–4.3%, respectively, with significant differences observed for the Ca concentration. In contrast, measurements of three Narutaki-to stones yielded concentrations of Fe, Si, and Ca in the ranges of 2.8–3.2%, 39–40%, and 0.03–0.05%, respectively, indicating high levels of Si. From these results, we find that the elemental concentrations of whetstones vary depending on the type of stone.7) We find that the concentrations of trace elements are the key to distinguishing different types of stone, and moreover that trace elements may be scattered non-uniformly over different regions of a sample. For these reasons, we used an energy-dispersive X-ray fluorescence analyzer to perform mapping measurements of regions of Ø1 mm. Our results indicated that the distributions of Al and Si were similar, and that Fe and Mn were abundant in several specific places: in the brown and black regions of Narutaki-to stones known as the “skin” or “contact” regions, in the purple gray regions that dot the surface of Binsui-do stones, and in the “gold stripe” regions of Uchigumori-do stones. We see that the results of fluorescence X-ray analysis provide information regarding the style and characteristics of naturally-excavated sharpening stones.8)

Fig.7 Results of measurements of natural whetstones
We have presented illustrative examples of elemental analyses of silver coins, copper coins, and natural whetstones. The results of elemental analysis, taken together with position information, are related to the characteristics of samples and may be considered an effective means of identifying the type and production site of excavated samples and archaeological resources. Indeed, in conjunction with the reconstruction of the Inariyama burial-mound sword excavated in Japan’s Saitama prefecture, which was completed last July, a whetstone excavated from Inariyama was analyzed. The results obtained for the Fe concentration suggested that the stone was of either the Aoto or Tsushima-do variety, with results for the Ca concentration indicating that Aoto was highly likely. This result, together with an investigation of sharpening residue on the surfaces of characters inscribed on the sword, led modern-day sharpener Okisato Fujishiro to conclude that the sharpening stone was an Aoto stone used in the final stage of the sharpening process. Similarly, a fluorescence X-ray elemental analysis combined with the results of mapping measurements proved useful in classifying and studying a Narutaki-to stone excavated near Tameike.8) It is important to make effective use of the results of non-destructive and non-contact analyses by working to establish correlations with detailed information obtained by destructive analyses. To this end, we can look forward to analytical instruments offering even greater precision and reduced measurement times.
The research reported here was a joint study with Keiji Umezaki, who provided copper coin samples; Yasushi Nishiwaki, who provided silver coin samples; and Okisato Fujishiro and Manami Takaoka, who provided whetstone samples. The research was conducted by several students in our university laboratory. Hitachi High-Tech Science Corporation assisted in developing dissolution methods for silver coin samples. We extend our gratitude to all of the contributors.
References
See more