
Kenichi Kobayashi
Ph.D. in Science
Assistant Professor, Medicinal Chemistry Department
Graduate School of Pharmaceutical Sciences
Meiji Pharmaceutical University
Instrumental analysis techniques for identifying the structures of organic compounds have made remarkable advances, to the point where ultra-small sample volumes suffice to allow structural determination even for complicated natural products with multiple asymmetric centers. With the exception of cases in which X-ray crystal analysis may be used to obtain unambiguous structures, structural determination for naturally occurring and synthetic organic compounds typically proceeds by amassing various types of instrumental data—such as NMR, IR, MS, specific optical rotation, and melting point data—and then attempting to draw conclusions by aggregating all available input. When submitting academic research articles in the field of organic chemistry, submission of the various instrumental analysis data is also mandatory.
Among these various types of instrumental analysis, X-ray crystallography, NMR, and MS occupy positions of particularly commanding authority regarding the structural determination of organic compounds, regardless of whether one is interested in known or unknown substances. X-ray crystal analysis is one of the most powerful tools available for identifying the structure of compounds, but cannot be applied in cases where the sample cannot be crystallized (although the recently reported crystal sponge method is a method of X-ray crystal analysis that does not require crystallization of samples)1). Similarly, NMR technology has developed rapidly in recent years and offers an enormous amount of information regarding the structure of compounds. However, in the structural determination of, say, a complicated natural product, the very complexity of the spectra frequently leads to proposals of incorrect structures based on erroneous analyses2). In contrast, mass spectrometry (MS) allows direct acquisition of molecular weight information for a compound. If fragment ions can be detected, one can obtain not only molecular weights but also partial structural analyses of compounds; moreover, application of high-resolution mass spectrometry (HRMS) can determine, among other properties, the atomic composition of a compound. For these reasons, MS is one of the most reliable and generally applicable techniques of instrumental analysis for identifying the structures of organic compounds.
In this work, we introduce a Hitachi High-Tech Science Chromaster 5610 mass detector in our laboratory and describe our use of this instrument to obtain mass information on synthetic organic compounds.
The Hitachi High-Tech Science Chromaster 5610 mass detector was developed on the basis of a new conceptual framework that differs from that of traditional mass analyzers. By combining this detector with a syringe pump, sample solutions of compounds of interest may be introduced directly into the mass detector to obtain mass information on the compounds. This allows determination of mass information on synthetic organic compounds as well as simple monitoring to verify compounds themselves when investigating reaction conditions.
When considering the installation of a mass analyzer in a university laboratory, a number of problems arise : where to install the device, how to manage the cost, and how to address questions of operation and maintenance (as students are wont to use equipment in rough ways). However, this product eliminates many of these concerns : its highly compact design gives it a reduced footprint on par with that of an LC system, and it does not require any complicated operating procedures. Its routine maintenance has been reduced to an extremely straightforward procedure: one need only to remove the atmospheric ion filter and subject it to ultrasonic cleaning. Moreover, the Chromaster 5610 is inexpensive compared to products from other vendors that offer similar functionality, a major advantage for installation in university laboratories.

Fig.1 The Chromaster 5610 mass detector
Our research group studies total synthesis of natural products for applications to the design of new anti-cancer drugs with a novel mechanism of action. We are working on a total synthesis of phaeosphaeride A, a naturally-occurring compound that is active in inhibiting STAT3 (signal transducer and activator of transcription 3); we have succeeded in the total synthesis of enantiomer of natural phaeosphaeride A3,4). In this work, we use the Chromaster 5610 mass detector to perform mass analysis of four synthetic intermediates according to the scheme shown in Figure 2.
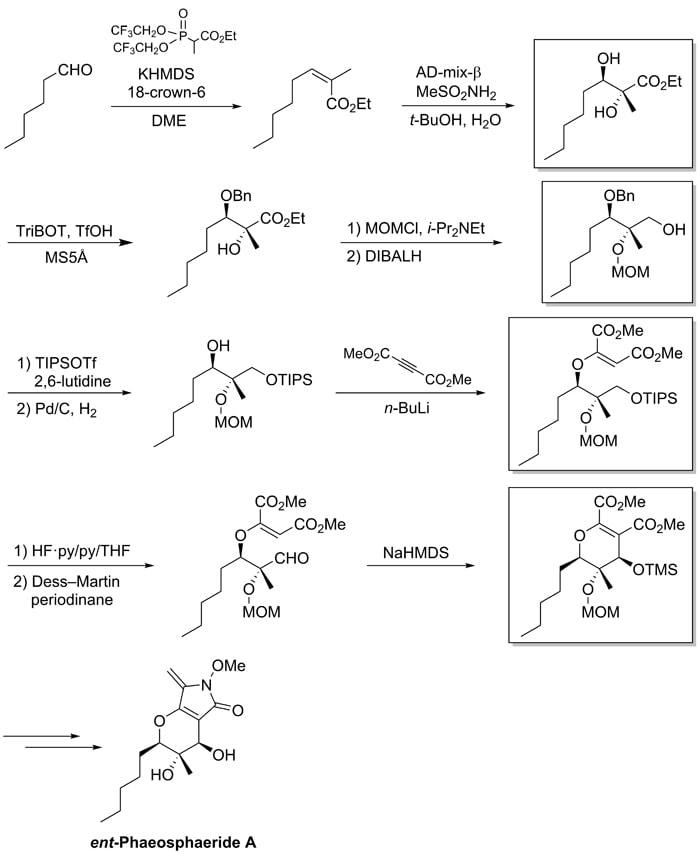
Fig.2 Synthetic scheme for ent-Phaeosphaeride A
Measurement conditions
Measurement conditions
Each of the compounds A-D is diluted to 10 ppm in methanol, then directly measured in the mass detector. Results are shown below. For each compound, we observe [M+Na]+, i.e., sodium-added molecular ions. In this way, we were able to obtain mass information on all compounds in just 5 minutes.
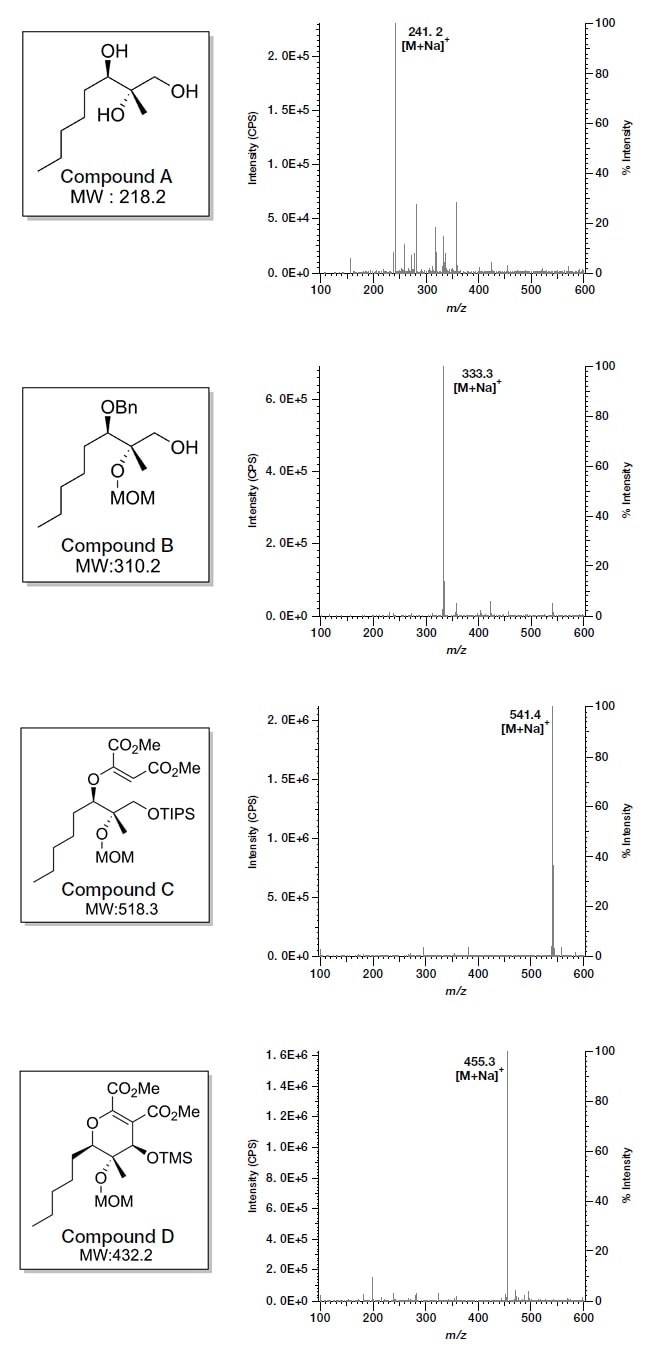
Fig.3 Mass information on synthetic compounds A-D obtained with the Chromaster 5610
In this work, we used the Hitachi High-Tech Science Chromaster 5610 to obtain mass information on synthetic intermediates arising in studies of the total synthesis of natural product. Although the samples measured in this work were small-molecule organic compounds with molecular masses in the range 200-500, the Chromaster 5610 is able to measure the masses of compounds with molecular masses up to around 1,000. The instrument boasts a small footprint, ease of use, and low cost, and promises to make major contributions to simple mass analysis at the laboratory level.
Acknowledgements
This research was conducted with assistance from a Scientific Research Grant, a Grant-in-Aid for Young Scientists (B) (Grant Number 25860015) from the Japan Society for the Promotion of Science. We are deeply grateful for this support.
References
See more