Sophisticated electron microscopy techniques successfully capture the world's first images
Honorary Professor Yoshinori Ohsumi of the Tokyo Institute of Technology received the Nobel Prize in Physiology or Medicine in 2016.
Dr. Misuzu Baba has played a key role in supporting the research that led to this achievement.
We have always wondered how Dr. Baba developed, among several other innovations, the ingenious electron microscopy technology that pioneered her novel yeast-specific rapid freezing and freeze-substitution method. We are also keen to know as to how she acquired her seemingly boundless passion for electron microscopy.
To learn more, we visited Dr. Baba's laboratory at the Hachioji campus of Kogakuin University, Japan.
Misuzu Baba, Researcher, Ph.D., Research Institute for Science and Technology, Kogakuin University

The 2016 Nobel Prize in Physiology or Medicine awarded to Honorary Professor Yoshinori Ohsumi of the Tokyo Institute of Technology for his contributions to "elucidating mechanisms underlying autophagy" cast a spotlight on the work of Dr. Misuzu Baba—a key supporting player in the research that led to the honor. Indeed, electron microscopy techniques developed by Dr. Baba provided the initial spark that fueled Professor Ohsumi's research on autophagy.
Dr. Baba first encountered electron microscopes while she was a student at Japan Women's University (JWU). As a second-year undergraduate in the Department of Biology, Faculty of Home Economics—the precursor of what is now the Faculty of Science—she attended a course on electron microscopy taught by Professor Masako Osumi. "We used an electron microscope to observe animal cells, and I was totally astonished—so this is what it actually looks like inside a cell! I remember thinking it was beautiful, and that the whole process was fun—to put it simply, I was moved."

Motivated by this experience, Dr. Baba found a copy of "Electron Microscopy: Illustrating the Cell", a 1974 book by Kazuo Ogawa and colleagues, and devoured the book from cover to cover. The electron microscope images of animals, insects, microorganisms, and bacteria illustrated in this book—albeit only in black and white—clearly revealed even the ultrastructure that are otherwise invisible to the naked eye, bringing to life the beauty of the microscopic world.
In her third year of undergraduate studies, Dr. Baba wanted nothing more than to try her hand at electron microscopy; she requested an assistant for an opportunity to make sections of the plant cells. "I was still a student", she explains, "so it was incredibly difficult to cut ultrathin sections, but somehow I succeeded and even got to observe my samples under the electron microscope. The first time I looked at a sample that I had cut, I saw a many scratch marks on sections, but I remember thinking that it was really fun to be able to see the intracellular structure of a plant cell wall." After that, Dr. Baba says, "I had no misgivings about diving headlong into the world of electron microscopy." JWU had an electron microscopy laboratory where Dr. Baba worked as a part-time assistant and as a research staffer in a biology course, eventually coming to use electron microscopes every day. "The extremely fortunate thing was that the equipment for electron microscopy was already in place at that time," Dr. Baba recalls. "Not only did we have transmission and scanning electron microscopes (TEM and SEM) but also freeze replica machines and cryo-ultramicrotome. Whenever a new piece of equipment was installed, I would give it a try, and eventually, I learned several different techniques of my own."
The superior techniques that Dr. Baba developed were the fruit of her own hard work and diligence. Through determined and repeated practice, she honed her sample-preparation skills, mastering techniques, such as ultra-thin sectioning, freeze replication, and cryo-ultramicrotomy. Furthermore, seminars played a large part in shaping her understanding of the instruments. Once, she applied to attend a seminar only to discover that she and another researcher were the only participants. Ordinarily, this would be a rather perfunctory affair, but here, Dr. Baba says, she was given an opportunity to disassemble a lens tube over and over again. Dr. Baba had learned the theory behind lenses from Ryohei Honjin's 1968 treatise Introduction to Electron Microscopy for Medicine and Biology. Also, whenever instruments in the electron microscopy lab required maintenance, she would watch over the technician's shoulder to observe what was happening, asking questions about the process and receiving lessons in instrument repair. Attending conferences, in which she learned new techniques, was a frequent occurrence. Not only was Dr. Baba skillful but she also possessed exceptional insight and was a quick learner. These qualities in her were later fruitful into the development of her extraordinarily powerful techniques in electron microscopy.
Of all the ingenious electron microscopy techniques Dr. Baba went on to develop, perhaps the most famous is the yeast-specific rapid freezing and freeze-substitution method. Dr. Baba had been using electron microscopy to elucidate the fine cellular structures of yeast—a fungus at Professor Masako Osumi lab in JWU. At that time, the most difficult problem in the study of the microscopic cellular structure of fungi was the challenge of fixation. Unless one succeeded in instantaneously stopping the metabolism of a live fungal cell and fixing it in place to prepare a sample, it was impossible to observe the dynamic state of organelles and other bodies within cells. "For animal cells," Dr. Baba recalls, "Professor Ichikawa and colleagues had been using rapid freezing methods for nearly 10 years to acquire huge numbers of electron microscope images. But for fungal cells like yeast, the cell wall always got in the way. Even with the same Heuser-style metal-contact approach used for animal cells, we never obtained good results. This gave me the sense that unless we damaged the cell wall—even if just slightly—the fixing fluid would never get into the cell and we had absolutely no hope of succeeding."
Around the same time, Professor Kenji Tanaka (then a professor at the Research Institute for Disease Mechanism and Control at Nagoya University School of Medicine) as well as the leaders of other research groups studying yeast cells were beginning to realize that rapid freezing would be necessary not only for animal cells but also for yeast cells. They contacted R. J. Howard and J. R. Aist, who had successfully applied rapid freezing and substitution methods to filamentous fungi belonging to the same family as yeast, and invited one of their researchers to deliver a seminar in Japan. Professor Tanaka and Dr. Hiromi Kobori—a researcher at JWU—attended the seminar to learn specific details of the methods. Nonetheless, success with yeast cells—whose cell wall exhibit properties distinct from those of filamentous fungi, making them difficult to handle—remained elusive.

"That was when I had the idea of using a sandwich structure to split the cell wall," Dr. Baba explains. "I sandwiched yeast cells between two copper plates and subjected them to immersion freezing, then fractured the cell wall by pulling the assembly apart, flowing osmium tetroxide (OsO4) into the interior of the cells. After innumerable failed attempts, I eventually succeeded in establishing this method as a viable technique. This method allows imaging of both the ribosomes and the cell wall—and I can't explain how very thrilled I was when this produced clear images. After all, at a time when the only available options were rather drastic—such as dissolving the cell wall, thereby destroying the ribosomes—I had finally succeeded in inventing my own methodology and also published a paper describing this new technique."
This was the birth, in 1987, of the so-called "sandwich method" for yeast-specific rapid freezing and freeze-substitution. Although this approach facilitated imaging of yeast cells in their entirety, it required well-honed skills and expertise; indeed, by around 1990, only three researchers in the world had successfully used the method to produce electron microscope images of yeast cells. Needless to say, one of these was Dr. Baba herself, whose dogged perseverance led to the development of this powerful new tool.
As Dr. Baba continued striving to refine her electron microscopy techniques, she gradually began interested in the outside world. In 1988, she transferred a researcher at Kogakuin University, after which she began visiting the laboratory of Yoshinori Ohsumi—who had just become an assistant professor at The University of Tokyo, College of Arts and Sciences—at Komaba campus. Professor Ohsumi had previously visited Electron microscopy lab of JWU to request freeze replicas imaging of isolated vacuoles. Eventually, Professor Ohsumi and Dr. Baba began a research collaboration. "As I acquired more and more technical skills, I started to feel a little dissatisfied with my situation and that was when Professor Ohsumi told me that he was going to open the Komaba laboratory. When I first started visiting this lab, the two of us were in the middle of discussing revisions to a research publication on which we had collaborated. For me, personally, I originally started working with electron microscopes because I had something to prove, but then the future directions of my research began gradually to take shape, and I found both the microscopes and the research to be fascinating and thoroughly absorbing. In fact," Dr. Baba laughs, "I was so riveted with my research that I did not even notice when a nationally famous Japanese pop singer got married. I was so completely absorbed in my research that I barely noticed anything else."
Meanwhile, Professor Ohsumi had established a new research initiative: studying mass transport in yeast vacuoles. Up to this time, vacuoles had been dismissed as little more than intracellular "trash dumps," but noting that they contained the same decomposing enzymes as lysosomes, Professor Ohsumi predicted that vacuoles also participate in intracellular decomposition.
The optical microscope observation of yeast cells in a state of starvation revealed a curious phenomenon in which large numbers of round particles seemed to accumulate inside vacuoles. "But what sorts of structures did these particles have? With low-resolution optical microscopy, there was simply no way to find out. And that was when Professor Ohsumi said to me, ‘I would like you to take a look at this and figure out what it is.’ That one simple sentence marks the birth of research on autophagy."
However, at the time Professor Ohsumi was sharing laboratory space with Professor Isao Katsura (now the Director of Japan's National Institute of Genetics); each researcher occupied half of the cramped space, and conditions were such that if the optical microscope was installed in the lab then the cell-culture apparatus had to be moved into the corridor. Compared to the "space age" environment at JWU, Dr. Baba recalls, the Komaba lab was more reminiscent of the "stone age." "I have extremely vivid memories of Professor Ohsumi asking me to take a look at his sample," says Dr. Baba. "When you dissolve the yeast cell wall to obtain a protoplast, osmium tetroxide can substitute; this is what Professor Ohsumi had done, and when he said ‘I don't care how dirty it gets—I just want you to observe it under the electron microscope,’ I first tried using ordinary fixation methods. As expected, the internal structure of the vacuoles was pitch black, and that was when I realized that I had no choice but to use rapid freezing methods, so I made up my mind to do it. However, the Komaba lab lacked the cell-freezing equipment required for fixation, and so I drew a diagram and took it to a manufacturing company in Hongo (Bunkyo City, Tokyo), and said ‘Make me one of these, please.’"

When the little cell-freezing machine was ready, it had no temperature control, but with a skilled and knowledgeable operator it was perfectly adequate. However, even Dr. Baba found the process of rapidly freezing yeast cells extremely difficult. This was because particularly for yeast cells in the starvation state, the cell walls are thick and the volume of the vacuoles is large. "Unlike cytoplasm," Dr. Baba explains, "vacuoles have high water content, so it was as if I had been asked to observe particles floating in water. In such a sample, ice crystals were easily formed during freezing, but these would destroy the structures in the interior and produce inferior images. It was challenging to identify the ideal frozen state, and I tried and failed many number of times. There were a number of other issues that made the sample processing somewhat of a nightmare—for example, the problem of surface alignment between the sample and the knife and the fact that ultrathin sectioning require more delicate handling than do thicker samples."
Eventually all the hard work paid off, and Dr. Baba finally achieved acceptable rapid freezing of yeast. Then, in 1989, she captured the world's first electron microscope images of the autophagy phenomenon. "The pictures were really beautiful," she remembers. "The particles in the yeast were round and those images made clear that they were surrounded by a membrane and that they were derived from the cytoplasm. When Professor Ohsumi realized that it (the substance comprising the bodies of the particles) was cytoplasm, he said, ‘With this I can publish two papers!’"
In fact, images could only be successfully captured if the rate of cooling was kept below 10,000°C per second—a fact that, like other aspects of the process, many practitioners are unaware of despite using many sophisticated component technologies. It's not easy to take beautiful pictures.
Prior to achieving this dazzling triumph, Dr. Baba had spent hours holed up at Kogakuin University putting all her energy into capturing electron microscope images. The instrument she used was a Hitachi model H-500H high-resolution TEM. After capturing images, she would write a report in her own words and set off for the Komaba lab, where a discussion ensued. At times, she says, she didn't set foot in the Komaba laboratory for stretches of almost 3 months. During that time, Hitachi electron microscopes were proving extremely useful in all settings but at Kogakuin University. "The Nissei Sangyo Co., Ltd. arm of the Hitachi Group (now Hitachi High-Tech) set up a laboratory in Ichigaya (Chiyoda City, Tokyo), and I even borrowed an electron microscope from them—either the H-7000 or the H-7100," says Dr. Baba. "Such a loan to a product user would be unimaginable today. Naturally, I'm extremely grateful to have had the opportunity to use the electron microscope as much as I wanted for an entire day. One of the images I captured at that time was included as a picture of a control cell in the publication cited as the reason for the Nobel Prize."
Dr. Baba summarizes the role played by electron microscopy in autophagy research this way: "It's no overstatement to say that the door to autophagy research was first opened when we saw that cytoplasm. That was definitely the first step. The second step was the use of the sophisticated technique of rapid freezing and freeze-substitution method to make observations; this allowed us to clearly observe membrane dynamics and also led to the discovery of autophagosomes in yeast. We were able to defi nitively unravel the mechanism of entry into the vacuoles. Using biochemical methods, it was not possible to observe the process through which the autophagosome merged with the vacuole membrane to achieve entry. With electron microscopy, however, we could clearly pinpoint the instant of vacuole entry. This could only have been possible via electron microscopy. Additionally, the images were really so beautiful that other labs just couldn't keep up with us."
This clarified the framework of autophagy, wherein vacuoles and autophagosomes merge, decompose unnecessary proteins, and create the proteins essential for survival. Professor Ohsumi published a paper on this discovery 2 years later, and the Ohsumi laboratory has remained a world leader in autophagy research ever since. Dr. Baba herself was awarded her PhD in 1996 for her thesis entitled "Morphological research on the autophagic function of yeast."
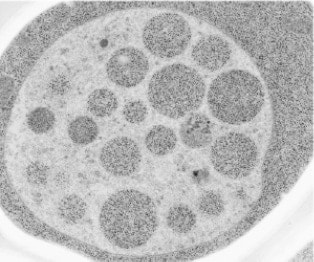
Incorporating autophagic bodies (cytoplasm) into the interior of vacuoles in nutrition-starved states.
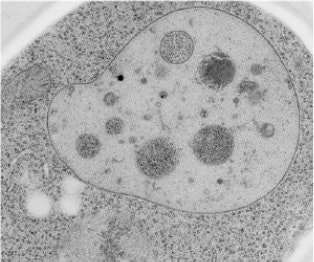
The incorporated cell after restoring vegetative growth.
The destruction of an autophagic body.
The awarding of the Nobel Prize to Professor Yoshinori Ohsumi on October 3, 2016, was simultaneously reported by many news outlets. However, Dr. Baba had assumed—given the extended streak of Nobel Prizes awarded to Japanese scientists—that "surely there won't be the ones this year" and had forgotten the matter entirely, right up until the day the prize was announced. A newspaper reporter placed a phone call to Professor Norio Baba—also a collaborator on autophagy research—so Dr. Misuzu Baba turned on the TV in a bit of a daze and saw Professor Ohsumi's press conference upon receiving the Prize.
"Of course, this is how research works", Dr. Baba muses. "He kindly mentioned my name fi rst during the conference. The prize was awarded to somebody I know so well and, while I was involved in the research being recognized, a part of me didn't feel like shouting ‘Hooray!’—instead, I was quietly awestruck and very, very grateful. After that I received some interview requests, and as time went on, it struck me more and more that this was a wonderful, and even a truly humbling, honor."
The autophagy research that began in 1988 has now been a part of both Professor Ohsumi's and Dr. Baba's lives for some 28 years. Today, Dr. Baba is pursuing collaborations with an overseas researcher who serves as the Chief Editor of an autophagy journal.
"I have experienced so many exciting moments—such as, if only I could see this one extra band!—that I am pretty sure I will never step away from autophagy research," says Dr. Baba. "From time to time, cells are kind enough to show us the decisive moments underlying the phenomena of life. So, for researchers, it is important that we not just stare blankly at what we're seeing—we must have the inquisitive eyes of an experimental scientist at all times. Of course, autophagy membranes are made for the specific purpose of being destroyed, so they are hard to detect. There was a time when I couldn't see the membranes clearly, and I was worried about that, but nobody was able to help me out. There was no clear path in front of me, but I knew I would be blazing a trail behind me as I went. That was the driving motivation for my work, and it remains my working philosophy to this day."
The phenomenon of autophagy—the recycling system into the cell—has been linked to Parkinson's and other neurodegenerative diseases and is increasingly expected to yield medical applications. However—in the words of Professor Yoshinori Ohsumi—we still understand no more than perhaps 30% of the phenomenon of autophagy. As Dr. Baba explains, "The origin of the membrane remains unknown, and our understanding of mutant strains is incomplete. There's a lot left to do."
Dr. Misuzu Baba gives every impression of refusing to rest until her electron microscope-powered morphological research on autophagy has resolved the last remaining mysteries in this domain of basic science. One careful step at a time.
(Interview and text: Ichiro Yamada)

Editor's note
There was no clear path in front of me, but I knew I'd be blazing a trail behind me as I went. Dr. Baba's candor and fearlessness have surely been inspirational to many readers. No matter what field of research, each and every scientist taking on the challenge of exploring the unknown is driven by a similar determination as they set off into an unmapped wilderness. With no precedent to serve as guidance, even minor roadblocks can create major confusion and yet the adventurers proceed—one careful step at a time. There is a word for the driving force that keeps them going—passion.
Mr. Hideaki Koizumi said it all in a previous issue (INTERVIEW Vol. 6, The Last Stage of Imaging): "If you have passion, then you just keep going until you break through—even if everyone and everything around you tries to keep you back. The trick is to put together a good team and set out with that mindset—that is how innovation happens." Dr. Baba's example proves that Mr. Koizumi has it exactly right.
This marks the 7th issue of the INTERVIEW series of special editions from SI NEWS. The researchers highlighted in each issue have inspired us with the intensity of their dedication and their commitment to persisting in the face of insurmountable obstacles—a vital wellspring of enthusiasm that furnishes the driving force to power through each day. At SI NEWS, our ongoing mission is to deliver that inspiration to our readers—reporting that makes you stop and double-check to make sure you didn't misread.
(Reporting: Tomoe Otsuka)
See more