
Toyohide Takeuchi
Professor, Doctor of Engineering
Department of Chemistry and Biomolecular Science, Faculty of Engineering
Gifu University
In previous work, the author and collaborators observed retention of hydrophobic anions (nitrate, iodide, and thiocyanate) via polyethylene glycol applied as a physical coating to hydrophobic stationary phases such as C30.1) Because the retention of sample anions increases as the concentration of the eluent rises, by using a high-concentration eluent we successfully measured quantities of iodide and thiocyanate ions in seawater.2) We believe that anions are retained by their distribution with respect to the polyethylene glycol phase. In this report, we describe efforts to develop chemically-bound stationary phases based on polyoxyethylene groups to improve the stability of the stationary phase.
This research was conducted in the hope of pioneering separation techniques based on novel mechanisms through the development of new stationary phases. As compared to conventional stationary phases such as C18, our stationary phases contain multiple oxygen atoms with an ether character, and we have high hopes for the effects produced by these oxygen atoms.
Aminopropyl silica (0.2 g) and a methyl polyethylene glycol N-hydroxysuccinimide (NHS) imide ester reagent (0.1 g) were added to a phosphate buffer solution (pH 7.0), allowed to rest for 30 minutes at room temperature, rinsed with water and methanol and then filled into a fused-silica capillary. Figure 1 shows the reaction scheme expected for this step.3) Because the NHS imide ester reagent used in this reaction is expensive, it is most useful for capillary LC measurements that require only small quantities of a stationary phase in the column.
The chemically-bound stationary phase of crown ether—a cyclic polyoxyethylene—was prepared as follows. Glycidyloxypropyltrimethoxysilane (0.2 mL) and dried toluene (1.2 mL) were added to silica gel (0.2 g) and allowed to react for 20 hours at 110 °C. After addition of the glycidyl group, 2-aminomethyl 18-crown-6-ether (60 mg) and dried toluene were added. The mixture was then allowed to react for 18 hours at 90 °C, and was rinsed with methanol and water. For the purposes of opening any remaining glycidyl-group rings, a 20% acetonitrile aqueous solution of 10 mM sulfuric acid was added, allowed to react for 4 hours at 40 °C, and was then filled into the fused-silica capillary. Figure 2 shows the reaction scheme expected for this step.4)
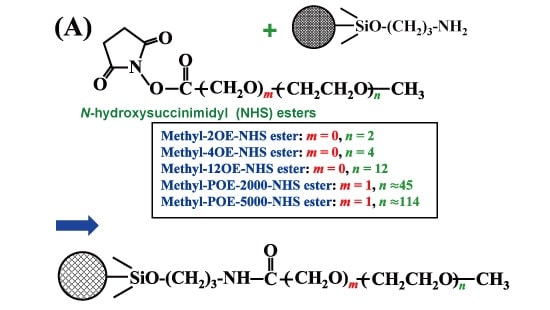
Fig.1 Preparation of polyoxyethylene stationary phase
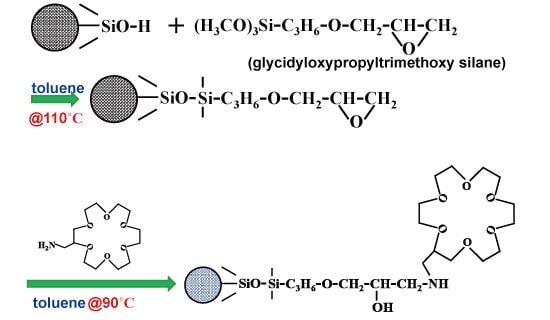
Fig.2 Preparation of crown-ether stationary phase
Using the stationary phase with bound polyoxyethylene groups, we found that the retention of anions decreased with increasing eluent concentration, and that it was possible to detect anions by indirect absorption. These findings demonstrate successful separation of anions in ion-exchange mode.5) The fact that this could be successfully achieved even using a stationary phase with an oxyethylene unit number equal to 2 suggests that cations in the eluent coordinate with multiple polyoxyethylene groups to function as anion-exchange sites. Figure 3 shows the results of a typical separation.3) Although we were not successful in separating nitrite and bromide ions, the order in which these species eluted was the same as that observed in ordinary ion-exchange mode. We were able to measure quantities of nitrate, iodide, and thiocyanate ions in saliva. In addition, we found that the polyoxyethylene stationary phase could be operated in HILIC mode by using high-concentration (70%) acetonitrile as an eluent.3) Moreover, when sodium dodecyl sulfate was added, sodium ions coordinated with polyoxyethylene groups and dodecyl sulfate group bind, as counterions, increasing the hydrophobicity of the stationary phase and demonstrating successful achievement of reversed-phase separation.3) These findings demonstrate that the separation mode can be selected by the choice of eluent for the polyoxyethylene stationary phase.
Meanwhile, the 18-crown-6-ether-bound stationary phase also exhibited crown ether complexation with cations in the eluent, suggesting that the trapped cations function as ion-exchange groups for anions.4) In this case, the extent of the retention varies depending on the type of cation in the eluent; cations that form more stable complexes with crown ethers tend to result in greater retention.
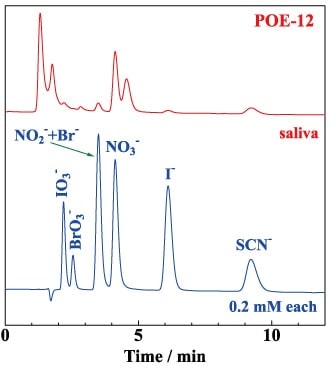
Column: POE-12, 100×0.53 mm
Eluent: 50 mM sodium sulfate
Flow rate: 8.0 µL/min
Samples: standard, 0.2 mM each; 10 times diluted saliva with deionized water
Wavelength of UV detection: 210 nm
Fig.3 Separation of anions by polyoxyethylene stationary phase
We expect that the polyoxyethylene stationary phases we have developed should find application to the separation and quantitative measurement of inorganic anions via ion-exchange mode and to the separation of polar compounds via HILIC mode.
References
See more