
Ryoji Kanno
Ph.D.
Professor
Director, Research Center for All-Solid-State Battery
Institute of Innovative Research
Tokyo Institute of Technology
Batteries are a hot topic in today’s world. Since their original invention by Volta over 200 years ago, many types of batteries have been developed and adopted for practical use, revolutionizing the world around us. The roster of useful battery architectures, which in earlier generations featured such stalwarts as lead-acid and zinc-carbon cells, was extended in 1991 by the arrival of lithium-ion batteries (LIBs), which are now widely deployed as power sources for devices such as video cameras, cellular terminals, smartphones, and even electric vehicles. Unfortunately, the reality is that today—some 30 years after the advent of lithium-ion batteries—we are only just beginning to understand how to use these devices in vehicles—a state of affairs that testifies to the agonizingly slow pace of progress in battery technology.
Just as lead-acid cells and similar workhorse devices furnished trustworthy foundations for daily life throughout the first 150 years of the battery era, lithium-ion batteries are widely expected to play a key role as energy-storage devices enabling the society of the future. But can this technology—which is already beginning to exhibit signs of approaching fundamental performance limits—truly remain viable throughout the next 50 to 100 years? The possibility that it may not motivates efforts to develop the next generation of energy-storage devices. Among the various candidate technologies that have been proposed for next-generation batteries, at Tokyo Tech’s Research Center for All-Solid- State Battery we focus on one particular approach: the all-solid-state battery, in which the electrolytic solution used in conventional lithium-ion batteries is replaced by a solid electrolyte, yielding a fully solid-state battery with no liquid components. The crucial challenge here is the design of the electrolyte itself, which must be a solid material through which ions can diffuse at high speeds. In 2011 we were thrilled to discover that lithium, phosphorus, sulfur, and germanium could be combined to yield a novel solid material—Li10GeP2S12—through which lithium ions move even more rapidly than through liquids at room temperature (Figure 1). We successfully demonstrated that all-solid-state batteries with solid electrolytes made from such materials not only achieve more than twice the output performance of conventional liquid-electrolyte LIBs, but also retain outstanding energy-storage properties over an extremely wide range of operating temperatures, from -30 to 100°C and above. The years since this breakthrough have seen vigorous efforts by researchers at many institutions to deploy all-solid-state batteries for practical applications.
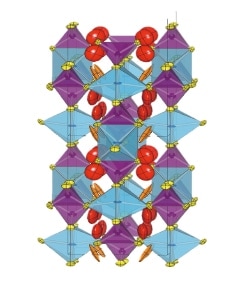
Fig. 1 Crystal structure of superionic conductor LGPS.
In the process of developing new solid-electrolyte materials, we discovered new crystal structures optimized for ionic conduction, with one-dimensional and twodimensional conduction pathways filled to capacity by a continuous distribution of lithium ions. Why such structures arise in practice is an extremely interesting question.
A battery is an amalgam of multiple materials, each playing a distinct role in realizing functional components such as positive electrodes, negative electrodes, and electrolytes. For this reason, the performance of batteries is directly tied to the quality of their constituent materials. However, although thorough understanding and control of constituent materials is a necessary condition for achieving high-performance batteries, it is far from sufficient. Indeed, the physical properties of individual materials and the ultimate performance of the battery are separated by an enormous conceptual gap that cannot be bridged with the present state of knowledge. It is this gap that I wish to emphasize here. A typical development sequence might be summarized as follows. The process begins with the creation of novel substances exhibiting new physical properties. This motivates a search for the optimal combination of materials to best exploit these new properties for maximal performance. One or more simple proof-of-concept devices are fabricated and tested to demonstrate basic principles of operation, after which begins a technology-development process to design practical devices. I will focus here on two complementary aspects of this process: the creation of novel substances and the creation of practical devices.
Creating novel substances. The creation of novel substances is just what it sounds like: the invention of new varieties of matter. Every era of human history has witnessed the advent of new materials whose innovative properties wield the destructive power to change the world. Needless to say, novel substances do not appear from out of nowhere. For synthesizing inorganic substances, there exists the helpful guidebook known as the periodic table, which acts as a repository for the accumulation of human wisdom and knowledge to date. The trick is somehow to extract the relevant pieces of information and combine them to yield desired substances—a challenge, of course, that we synthetic chemists are uniquely well-positioned to address. Using the full breadth of knowledge and experience we have accumulated to date, we combine elements via synthesis reactions—performed under conditions believed to be necessary—to yield new substances. Sometimes this goes well, and sometimes it doesn’t, and in either case our mandate as researchers is to come back the following day—and every day—and try something else. At each iteration of this trial-and-error process, we attempt—based on the knowledge and experience we have acquired thus far—to answer a simple question: Can such a substance truly exist? If not, how might we tweak the combination of ingredients or the synthesis conditions to nudge it closer to reality? This is the basic exploratory process through which our research progresses. In a sense, it is really nothing other than a dialogue with the putative target substance we are trying to create (or with someone attempting to will it into existence), and it is a dialogue that leaves absolutely no room for compromises, fudge factors, or sleight-of-hand. The capacity to enjoy this conversation with nature—while advancing research with a steady focus on the road ahead over the long term—is the key factor for success. The researchers who succeed in creating new substances, and transforming them into revolutionary new materials, are those who are patient—and passionately dedicated to the goal of creating something new. In the future, advances in computing power will unquestionably yield dramatic improvements in the power of modeling tools to assist in designing materials; nonetheless, with or without these tools the basic mindset needed to create new substances will remain unchanged.
Creating practical devices. The emergence of new substances with novel properties leads first to materials research—to study how the new properties can be exploited in devices—and later to more advanced device engineering. Here again the challenge of creating new devices requires commitment and dedication on the part of engineers: initial proof-of-concept experiments must be developed into practically useful devices, whose adoption requires overcoming resistance from users accustomed to conventional devices. Indeed, the more dramatic the advance achieved by a new device, the more strenuous the resistance to its adoption (cf. the phenomenon of creative destruction). The question of whether or not the world eventually accepts the device hinges on the resolve of the technology developers—and their wherewithal to shepherd their creation through obstacles. Extracting the greatest possible performance from a new device requires a comprehensive, all-hands-on-deck effort exploiting every last iota of existing knowledge—including not only scientific and technical insight, but also wisdom from various other disciplines required for accurate assessment of societal needs. Large engineering teams demand sophisticated management of the technology-development process, requiring intimate familiarity with government policy and other provisions required for institutions designed to interface with human counterparts. This, too, becomes possible only when many passionate individuals unite and work together to realize a shared dream.
Needless to say, the two complementary creation processes we have described—one pitting researchers against nature and god, and the other attempting to harness the collective power of distinct but like-minded individuals to advance technology development—often wind up at loggerheads. But the rare cases in which the two pursuits intermesh and progress together give rise to new technologies with the power to change the world. I am hopeful that solid-state battery technology will be history’s next example.