Atsuko Yamada
Modern mass-production, mass-consumption societies discard enormous quantities of unneeded waste products on a daily basis. Failure to process these waste products appropriately could pose grave environmental problems; we must therefore pursue the goals of reducing, reusing, and recycling waste. Figure 1 illustrates common processes for handling various types of waste. As this figure suggests, techniques for making effective use of waste products—and the analytical tools required by those techniques—differ depending on the type of waste and the relevant processes. In this article we focus specifically on ash and slag.
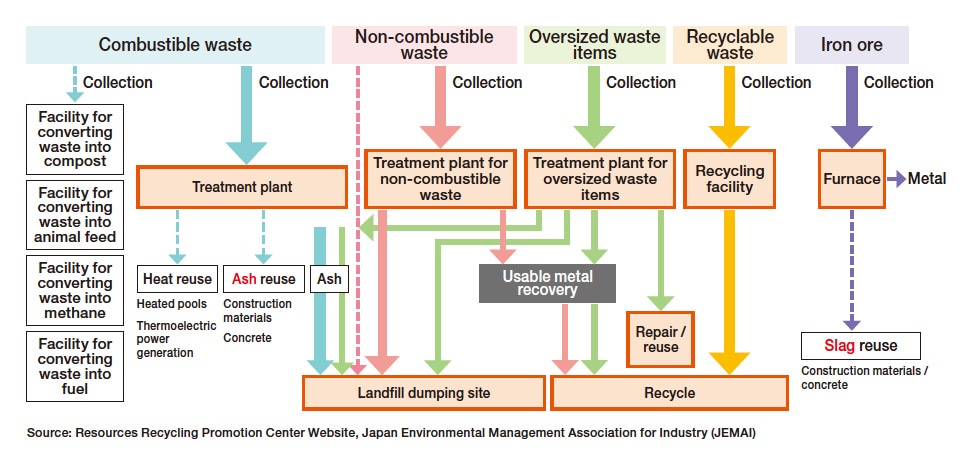
Fig. 1 Processes for handling waste products.
Ash and slag may be reused to produce concrete or other construction materials, but this requires analysis and maintenance procedures to verify that the material composition is suitable for this purpose and that toxic-element concentrations are below regulatory thresholds. Methods for performing these analyses are specified by two industrial standards: JISM8815:1976, "Methods for Analysis of Coal Ash and Coke Ash," and JISK0058-2:2005, "Test Methods for Chemicals in Slags, Part 2: Test Method for Acid Extractable Contents of Chemicals." However, the procedures described in these standards involve complicated operations and pre-processing steps, making them unsuitable for rapidly analyzing large numbers of test samples. To overcome this problem, screening methods based on energydispersive X-ray fluorescence analysis—which requires no complicated preprocessing, allows analysis of solid, liquid, and powder samples, and can perform non-destructive analysis and simultaneous multiple element measurement—are frequently used1).
In this study, we analyzed ash and slag using the EA1400 energy-dispersive X-ray fluorescence analyzer from Hitachi High-Tech Science. The EA1400 is equipped with an X-ray-emitting vacuum tube and a semiconductor detector positioned beneath the sample stub; after mounting a sample, a measurement lasting just a few minutes suffices to allow qualitative and quantitative analysis of the elements (from 11Na to 92U) contained in the sample. Boasting a new high-resolution, high-count-rate silicon drift detector (SDD) and vacuum system that enable higher-sensitivity, higher-throughput measurements than existing energy-dispersive X-ray fluorescence analyzers, the EA1400 is widely used for quality control and screening of regulated elements, and has also been employed for a broad range of other purposes, including environmental studies and analysis of cultural artifacts.

Fig. 2 Energy-dispersive X-ray fluorescence analyzer EA1400.
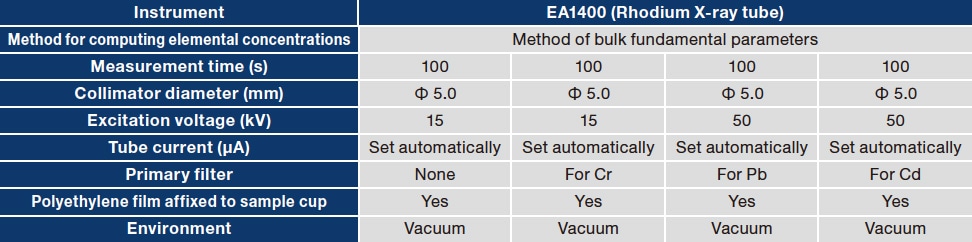
The ash sample we used was a reference sample of coal ash (certified reference material JSAC 0522) from the Japan Society of Analytical Chemistry. The slag sample was a water-granulated slag that we obtained independently and crushed to ensure particle diameters below 0.25 mm. We prepared two sample cups— of inner diameter 24 mm and height 22 mm with polyethylene films affixed to the lower surfaces—to which we added approximately 10g of each sample, and then covered with paper lids. We measured each sample using the EA1400; to increase sensitivity to fluorescence X-rays from elements with low atomic numbers (up to 20Ca), we used a rotary pump to evacuate the specimen chamber to a vacuum state and set the measurement time to 400 seconds.
Table 1 Experimental conditions used to measure samples of coal ash (certified reference material JSAC 0522) and water-granulated slag.
The measurement results obtained using the EA1400 are listed in Table 2. For the coal-ash sample we compared EA1400 measurement results to standard tabulated values for this certified reference material (JSAC 0522). For the slag sample we compare EA1400 measurement results to those obtained using an inductively-coupled-plasma optical-emission spectrometer (ICP-OES).
Table 2 Results of EA1400 measurements compared to standard tabulated values (for coal-ash sample) or to ICP-OES measurements (for slag sample).
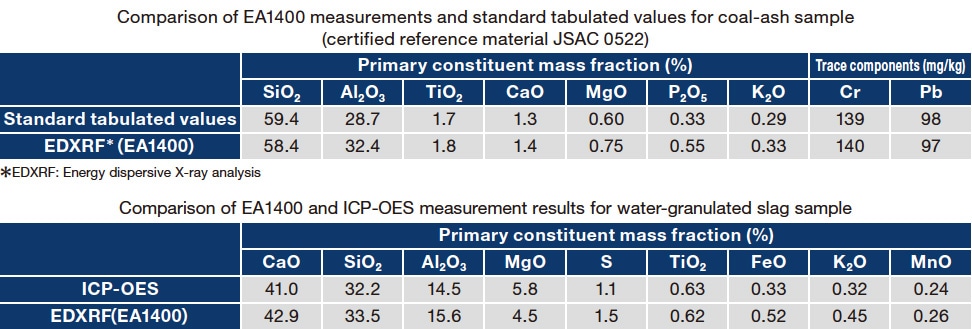
The EA1400 measurement results closely match standard tabulated values (for the coal-ash sample) or ICP-OES measurements (for the slag sample); we attribute this good agreement to the meticulous care we have taken to ensure satisfaction of the key requirements summarized in Table 3.
Table 3 Key requirements for successful X-ray fluorescence analysis

A first essential prerequisite for successful X-ray fluorescence analysis is to use observation samples of homogeneous composition; it was for this reason that we crushed the water-granulated slag to yield homogeneous slag samples for EA1400 measurements. The sample volume we chose (10 g) was designed to ensure that each sample would have thickness sufficient to constitute a bulk region. This is considered to be the case if varying the sample thickness does not affect its fluorescence X-ray intensity. The minimal thickness required depends on the primary constituents of the sample and on the fluorescence X-ray energies for the elements measured; for ash and slag samples, it is around 5mm.
In this study, we quantified elemental concentrations using the method of fundamental parameters (FP): I f t he composition of a sample (all elements contained in the sample and their relative abundances) is known, the expected intensities of fluorescence X-ray emissions may be computed theoretically—based on the principles of fluorescence X-ray emission—in terms of experimental parameters and physical constants (or “fundamental parameters.”) The FP method determines the composition of a sample by comparing these calculated theoretical intensities to experimentally measured emission intensities for the sample.
Although there are formulas for converting measured intensities directly to element abundances, these formulas are complicated and difficult to use; instead we use a method of successive approximations, which proceeds as follows. We begin by determining an initial estimate—a starting guess—for the sample composition, then compute the fluorescence X-ray intensities for this composition and compare them to the measured intensities. If the computed and measured intensities greatly disagree, we adjust our composition estimate appropriately, then re-compute the expected intensities and compare to measured intensities. Continuing in this way, we repeatedly adjust our composition estimate until the discrepancy between the computed and measured intensities is sufficiently small; the composition estimate for which computed and measured intensities agree is then returned as the result of the analysis2).
The primary elemental constituents of both ash and slag are known to exist in the form of oxides; thus, in this study we assume that the primary elements for which we measure fluorescence X-rays exist within samples in the form of their typical oxides. From Table 3 it can be seen that, by taking pains to ensure that all key requirements are satisfied, the EA1400-based analysis succeeds in determining accurate concentrations not only for the principal components of coal ash and water-granulated slag, but also for the trace components (Cr,Pb) present in coal ash. These results demonstrate that X-ray fluorescence analysis, despite being a highly sensitive tool, is nonetheless capable of yielding analytical results that closely match those obtained via the analytical procedures specified by industrial standards—and will likely prove a powerful technique for analyzing recycled materials.
Energy-dispersive X-ray fluorescence analysis requires no complicated preprocessing, allows analysis of solid, liquid, and powder samples, and non-destructive analysis and simultaneous multiple element measurement—and is thus a powerful analytical tool for screening recycled materials, an application demanding rapid analysis of large numbers of test samples. In this study we demonstrated that X-ray fluorescence analysis—with careful attention paid to ensuring the satisfaction of certain key requirements—can yield results similar to those obtained via the analytical procedures specified by industrial standards. We expect that X-ray fluorescence analysis, a technique already used in a broad variety of fields, will also become a widely-used tool for analyzing recycled materials.
References
About the authors
Atsuko Yamada
Application Development Center
Hitachi High-Tech Analysis Corporation
See more