
Kenta Kanosue
Doctoral Student
Department of Chemistry and Materials Science
Graduate School of Science and Engineering
Tokyo Institute of Technology
(Doctorate in Engineering expected March 2017)

Shinji Ando
Professor (Doctor in Engineering)
Department of Chemical Science and Engineering
Tokyo Institute of Technology
Polyimides (PIs) are polymers that have dense aggregation structures and highly ordered structures arising from the rigid chemical structure and strong intermolecular interactions. For this reason, PIs can be used to produce super engineering plastics, with properties such as high tolerance to high and low temperatures, outstanding mechanical characteristics, and the ability to withstand exposure to light and radioactivity for long periods of time; they are widely used in fields such as the semiconductor and aerospace industries.1) In recent years, polymer thin films with the ability to absorb ultraviolet light and emit visible light—wavelength-converting properties—have been a focus of intense interest, and have promising potential applications as wavelength-conversion films (downconverters).2-4) When positioned on the outermost surface of a commercial silicon-based solar cell, downconverters transform ultraviolet radiation—for which silicon has poor spectroscopic sensitivity—to the visible radiation, where the sensitivity is higher; this improves the total photoelectric conversion efficiency of the device. The practical ability to fabricate such wavelength converters from luminescent PIs offers the possibility of designing highly efficient and durable solar cells.
The essential luminescence properties required for downconverters are high transparency in the visible region, high luminescence quantum efficiency, and a large Stokes shift. Stokes shift is energy difference between the wavelengths of the absorption and emission peaks; the larger the Stokes shift, the higher the effi ciency with which ultraviolet radiation is converted to longer-wavelength visible or near-infrared light. In our recent studies targeting the development of novel PI thin films exhibiting these properties, we have focused on room-temperature phosphorescence. The highly fluorescent PI thin films that we have developed thus far exhibit relatively small fluorescence Stokes shifts; thus, in case that PIs are designed to give colorless, transparent films, they exhibit signifi cant emission only in the short-wavelength region of the visible spectrum (violet-blue), while for thin films designed to have signifi cant emission at longer wavelengths (green-red), yellow or red coloration of the film was unavoidable (Fig. 1).5,6) Unlike fluorescence, phosphorescence is emission from an excited triplet state lying at a low energy level that proceeds via an intersystem crossing, generally resulting in a large Stokes shift. In the past, it was believed that phosphorescence could only be observed for (a) heavy metals, rare-earth complexes, and other systems exploiting the effects of heavy atoms in organic compounds, or environments in which molecular motion is frozen or otherwise suppressed, such as (b) highly crystalline materials, or (c) systems at low temperatures, such as the temperature of liquid nitrogen (77 K). However, it has recently been reported that organic carbonyl compounds with heavy-halogen atoms such as bromine or iodine can exhibit phosphorescence near room temperature.7) In recent work, we have discovered that semi-aromatic PIs and low-molecular-weight imide compounds containing bromine or iodine in their dianhydride moiety can exhibit room-temperature phosphorescence with extremely large Stokes shifts (on the order of 10,000 cm).1),8) In the near future, the development of PI materials suitable for applications to solar-cell downconverters using room-temperature phosphorescence will require characterization of phosphorescence properties in a variety of environments chosen to focus on practical applications. In particular, it is known that phosphorescence depends strongly on temperature and oxygen concentration in the measurement environment, so that analysis of phosphorescence at variable temperatures and in oxygen-free environments is essential.
In this article we present the results of measurements at low temperatures and in vacuum of emission spectra from two types of materials which we have developed: a low-molecular-weight phosphorescent imide compound (powder) and a PI thin film formed on a substrate.
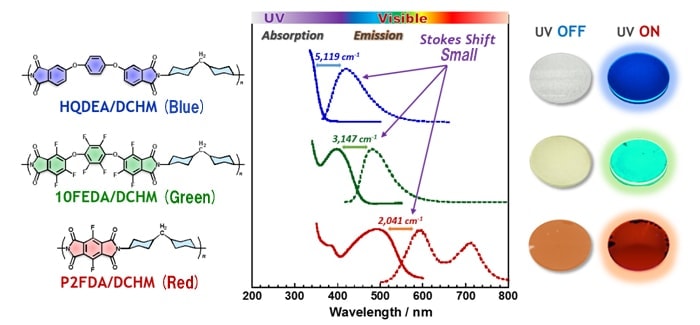
Fig 1.
Highly fluorescent polyimides (PIs): molecular structures, UV-visible absorption and fluorescence spectra for PI thin films, and images of PI thin films in the absence and in the presence of ultraviolet irradiation, exhibiting different fluorescence colors.
A F-7100 fluorescence spectrophotometer (Hitachi High-Tech Science) was used to measure emission spectra of the samples. For measuring emission spectra at variable temperatures, sample holder for solid fluorescent samples (Unisoku CS-KF-H07-42) was mounted on a compact temperature controller (Unisoku CoolSpek UV USP-203-B) and was installed in the sample chamber (Fig. 2a). Liquid nitrogen was used to cool the sample and the sample chamber. By circulating nitrogen gas in the vicinity of the sample chamber, cooling could take place without the need to immerse the sample in liquid nitrogen. Measurements were taken at temperatures in the range -175 to +100 °C. To prevent misting of the sample chamber interior and of the inner and outer surfaces of fused quartz window, the apparatus was designed such that the interior of the sample chamber was nitrogen-exchanged and its exterior was heated by instrument's built-in heater. To measure emission spectra in vacuum, a vacuum chamber (Akada Kogyo) designed to the specifications of our laboratory was installed in the F-7100 sample chamber (Fig. 2b), and measurements were performed after pumping out the interior of the chamber for 1 hour with a vacuum pump (PFEIFFER XtraDry 150-2).

Fig 2.
(a) Apparatus for measuring emission spectra at variable temperatures (Unisoku)
(b) Apparatus for measuring emission spectra in vacuum (manufactured by Akada Kogyo to Ando Laboratory specifications)
Figure 3 shows the emission spectra, measured at room temperature under atmospheric pressure, for a phosphorescent imide compound in the solid state (powder) and for a phosphorescent polyimide (PI) thin film. The imide compound and PI thin film were excited by UV light at 330 and 315 nm, respectively, and were observed to exhibit green room-temperature phosphorescence at 510-530 nm, corresponding to extremely large Stokes shifts of ν >10,000 cm-1. Emission peaks originating from fluorescence were also observed near 450 nm for the imide compound and near 400 nm for the PI thin film. The positions of both the fluorescence and room-temperature phosphorescence peaks for the imide compound were red-shifted compared to those for the PI thin film. We attribute this to the energetically stabilized excited state in the imide compound, which is due to the effects of strong intermolecular interactions arising from the dense molecular packing in imide compound microcrystals. The luminescence quantum yield was estimated to be approximately 2% for both the imide compound and the PI thin film. In general, at room temperature and atmospheric pressure, local molecular motion—even in the solid (crystalline) state—can result in nonradiative deactivation or efficient energy transfer to oxygen molecules during the long lifetime of the excited triplet state, making highly efficient phosphorescence difficult to achieve. Thus, for the purpose of constructing molecular design guidelines to facilitate the future development of highly efficient phosphorescent PI thin films, it is important to study in detail the effect of molecular motion and oxygen on the phosphorescence properties.
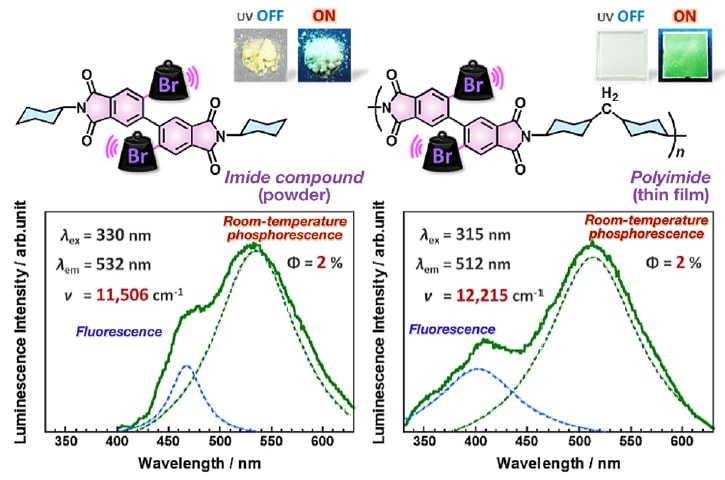
Fig 3.
Top: Images of phosphorescent imide compound (powder) and PI thin film with and without UV irradiation.
Bottom: Emission spectra of the imide compound and PI thin film measured at room temperature and atmospheric pressure.
Local molecular motion in organic compounds and polymer materials varies signifi cantly with temperature, and thus the effect of molecular motion on the phosphorescence properties could be investigated by measuring emission spectra from high temperatures to low temperatures. Figure 4 shows the emission spectra measured at various temperatures for two samples known to exhibit room-temperature phosphorescence: an imide compound (powder) and a PI thin film formed on a fused silica substrate. The temperature was varied from +100 to -175°C in steps of 25°C and the emission spectra were measured at each temperature. At 100°C, non-radiative deactivation due to the vigorous motion of highly activated molecules causes that almost no phosphorescence is observed. On the other hand, at low temperatures the suppression of molecular motion gives rise to a high phosphorescence intensity for both samples; for the imide compound the intensity at -175°C is approximately 26 times as high as the room-temperature value, while for the PI thin film the intensity at -175°C is approximately 46 times as high as the room-temperature value. For the PI thin film, the luminescence quantum yield at liquid-nitrogen temperature is 76%, an extremely high value. At temperatures near -175°C, we may assume that molecular motion in both samples is essentially frozen out, so the increase in phosphorescence intensity due to cooling could be understood as an indirect reflection of local molecular motion at room temperature. Imide compounds, due to crystallization at the time of preparation, exist in a crystalline phase with a three-dimensional ordered structure, and thus we may expect that the local molecular motion in these materials to be fairly well suppressed even at room temperature. In contrast, PI thin films form amorphous phase, and PI chains are entangled in complex ways to increase the free volume, and thus the local mobility of these chains is relatively high at room temperature. This is the most likely explanation for our finding that the increase in phosphorescence intensity upon cooling is relatively larger for the PI thin film. For both samples, the phosphorescence peak is blue-shifted from around 510-530 nm at room temperature to around 500 nm at -175°C. This may be attributed to the fact that molecular motion is strongly suppressed at lower temperatures, reducing the probability of geometrical stabilization (internal conversion) due to structural relaxation in the excited state. On the other hand, the fluorescence observed at 400-450 nm does not exhibit a prominent increase in intensity at lower temperatures, indicating that fluorescent emission is not sensitive to molecular motion. This is due to the extremely short lifetime of the excited singlet state which generates fluorescence.
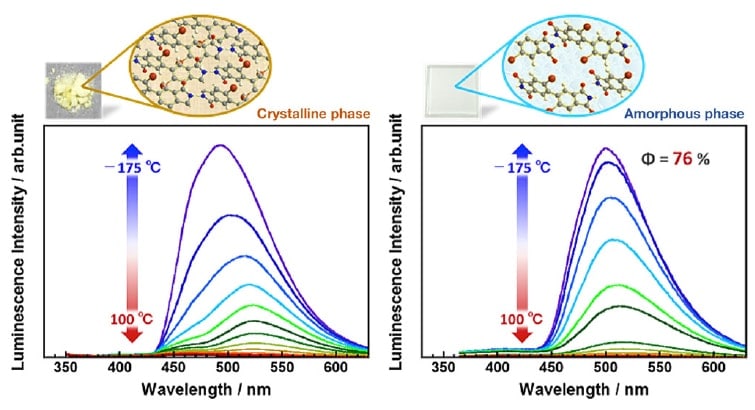
Fig 4.
Schematic depiction of condensation of molecular chains (insets) and emission spectra at variable temperatures (plots) for phosphorescent imide compound (powder, polycrystals) and amorphous PI thin film.
In general, phosphorescence is effectively quenched by oxygen molecules, for which the ground state has a triplet configuration. Thus, to investigate the effect of oxygen molecules on the phosphorescence properties of PI thin film, emission spectra were measured in vacuum. Figure 5 shows the emission spectra for a phosphorescent PI thin film at room temperature/atmospheric pressure and in vacuum. The room-temperature phosphorescence peak observed near 520 nm under atmospheric pressure increases in intensity by roughly a factor of 2 in vacuum. This indicates that, at atmospheric pressure, the phosphorescence efficiency is suppressed by energy transfer to oxygen molecules present at the surface and in the interior of the PI thin film. On the other hand, because energy transfer to oxygen molecules does not arise in the fluorescence process responsible for the peak observed near 400 nm, the intensity of this peak remains unchanged in vacuum.
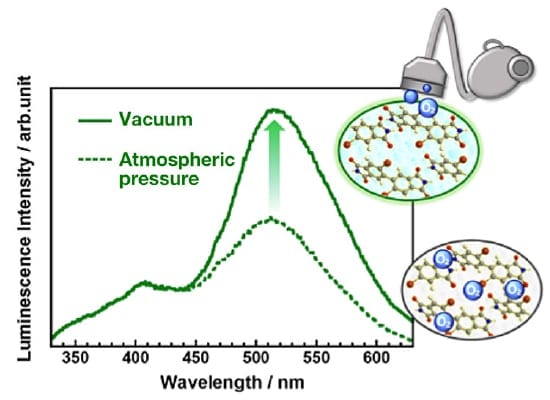
Fig 5.
Emission spectra for phosphorescent PI thin film at atmospheric pressure and in vacuum.
Only the phosphorescence peak increases in intensity in vacuum.
In general, phosphorescence is associated with long luminescence lifetimes on the order of microseconds or milliseconds. To investigate the effect of oxygen molecules on the phosphorescence lifetime of the PI thin film, phosphorescence spectra were obtained and phosphorescence short-lifetime measurements were performed in vacuum. To measure t he phosphorescence spectra, the built-in phosphorescence mode of F-7100 was used. This mode uses a 40 Hz optical chopper to convert a continuous beam from a light source into an effective pulsed light source with a pulse duration on the order of 2 ms. The sample was illuminated with this pulsed source, while simultaneously applying a delay of roughly 1 ms to the detector following the cutoff of each light excitation pulse, allowing detection of only phosphorescence and other light components after the fluorescence has been totally decayed (Fig. 6). The phosphorescence short-lifetime measurements were also performed using the built-in time-varying phosphorescence measurement mode of F-7100 spectrophotometer. In this mode, after a delay time of roughly 1 ms to allow the fluorescence to relax completely, data is acquired for roughly 23 ms and the decay of the phosphorescence is measured over 20 ms. The excitation and emission wavelengths were set at 315 and 515 nm, respectively, and measurements were made of phosphorescence lifetimes in the range 0-20 ms. Figure 7 shows the phosphorescence spectra for the PI thin film measured at atmospheric pressure and in vacuum. At atmospheric pressure, no clear peaks are observed in the three-dimensional phosphorescence spectrum, nor is an emission peak from the 315 nm excitation present. In contrast, in vacuum, a clear phosphorescence peak due to the 315 nm excitation is observed in the range 500-600 nm. Figure 8 shows the results of phosphorescence short-lifetime measurements for the PI thin film under atmospheric pressure and vacuum. At atmospheric pressure, no components with emission lifetimes on the order of milliseconds are observed, whereas in vacuum, a decay with a lifetime of ca. 20 ms is observed, with a profile typically characteristic to phosphorescence. The fact that an oxygen environment signifi cantly reduces the phosphorescence lifetime confirms that the luminescent PI we have developed indeed exhibits room-temperature phosphorescence.
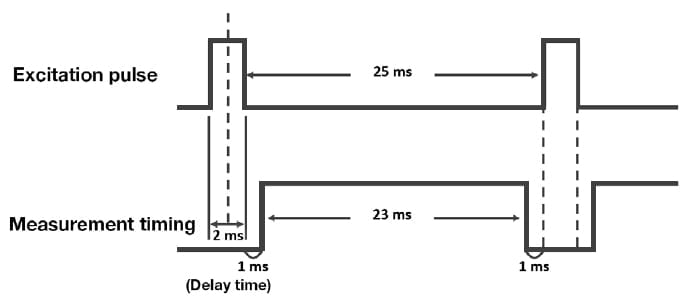
Fig 6.
Timing chart for phosphorescence measurement mode
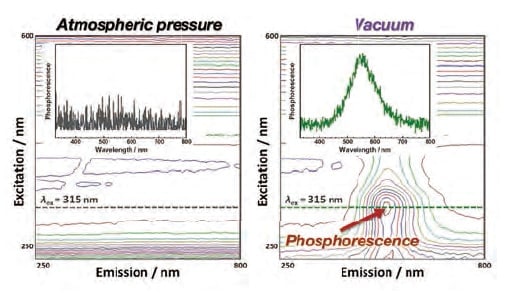
Fig 7.
Three-dimensional phosphorescence spectra for phosphorescent PI thin film at atmospheric pressure and in vacuum.
The insets show spectra for a fixed excitation wavelength of 315 nm.
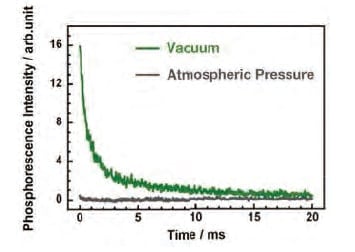
Fig 8.
Results of phosphorescence short-lifetime measurements for PI thin film at atmospheric pressure and in vacuum.
In this article we presented the results of measurements and analyses of excitation and emission spectra, at varying temperatures and under vacuum, for a room-temperature phosphorescent imide compound and a polyimide, with the objective of elucidating their characteristic phosphorescence properties. The temperature dependence of the emission spectra clearly demonstrated that differences in room-temperature molecular motion arising from the different three-dimensional and ordering structures of the imide compound (powder) and PI thin film have a strong influence on the phosphorescence properties. The emission spectrum of the PI thin film measured in vacuum revealed that oxygen molecules, present at the surface or in the interior of the film, quench the phosphorescence. The fact that the luminescence properties of the phosphorescent PI thin film depend sensitively on the temperature and oxygen concentration suggests that this material may find applications not only as wavelength-conversion films (downconverters) but also for temperature and oxygen sensing. Moreover, the fact that this material simultaneously exhibits strong resistance to high and low temperatures, illumination and harsh environments, and has the ability to emit strong luminescence at low temperatures and in vacuum, suggests potential applications to downconverters suitable for use in space. In future work, we plan to continue improving the wavelength conversion effi ciency of these materials.as well as their ability to withstand harsh climates and illumination—with an eye toward applications as sensing materials and for use in space.
References
See more