
Hiroshi Yamamura
PhD (Engineering)
Associate Professor
Department of Integrated Science and
Engineering for Sustainable Society
Faculty of Science and Engineering
Chuo University
Rivers, lakes, and other natural bodies of water contain many varieties of organic matter—but where does this organic matter come from? Sources of organic matter emissions may be broadly divided into two categories: animal-based and plant-based. The former category encompasses emissions from the vicinity of the bodies of animals—including liquid waste produced in daily life such as feces and urine, and dead bodies—with decomposing organisms constituting the primary emission sources. On the other hand, the primary sources of plant-based emissions are components emitted in conjunction with the decomposition of plant matter. Both animal-based and plant-based sources release organic matter in the form of large molecules with weights of a few million Da (the Dalton is a unit of molecular weight). However, a variety of mechanisms—including exposure to sunlight and the activity of microorganisms—induce physical, chemical, and/or biological oxidization and decomposition processes that reduce these large molecules to smaller molecules with weights of a few thousand Da. Through these natural decomposition processes, hydrophilic macromolecular organic matter originating from compounds such as proteins and polysaccharides experience an increased degree of unsaturation (carbon-carbon double and triple bonds), significantly shifting their chemical properties towards those of highly hydrophobic brown-colored organic matter.
The highly unsaturated, several-thousand-Da organic matter produced by decomposition is known as humic substances, and, in a sense, most naturally occurring organic matter is composed primarily of humic substances. Humic substances are a primary source of organic matter in soil; in addition, their tendency to form complexes with metals ensures that humic substances play an important role in cycling matter through the natural world, facilitating, for example, the transport of iron from forests to oceans. On the other hand, in water-treatment processes for tap water, it is known that chlorine, added as a disinfectant, reacts with humic substances to form the carcinogen trihalomethane. In some regions with high concentrations of humic substances it is therefore necessary to carry out a detoxification step in which these substances are removed by ion exchange or other methods. In summary, insight into the behavior and properties of humic substances provides important input for both natural sciences and engineering disciplines. Research studies on the abundance of humic substances and their physical and chemical properties have been actively pursued at bodies of water around the world, from the South Pole to the heart of the Amazon1).
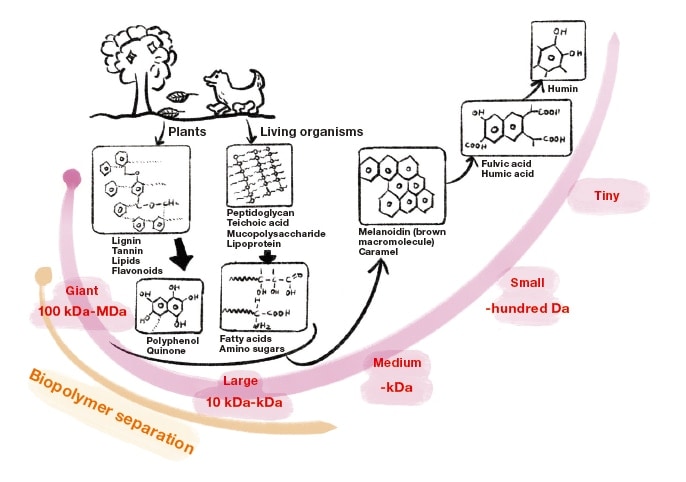
Fig. 1 Process of organic-substance decomposition in natural bodies of water.
Because "humic substances" is a general term describing all substances produced by the decomposition of organic matter in the natural world, it encompasses a wide range of matter spanning a broad range of molecular weights, whose constituent elements, moreover, vary extensively depending on the season and the body of water in question2). In addition, the extremely complicated chemical structures of these compounds defy efforts to define their chemical composition in a uniform manner; instead, one typically uses methods such as chromatography to separate substances on the basis of similar physical or chemical properties before proceeding to qualitative and quantitative analysis. A particularly useful technique for this purpose is high-performance size-exclusion chromatography (HPSEC), which is capable of simultaneously separating humic substances spanning a vast range of molecular weights, from a few thousand to a few million Da, into multiple categories, all in a single experiment. The remarkable separatory power of this method has led to its use for many years to analyze organic matter in natural bodies of water. Prior to the year 2000, HPSEC instruments commonly employed ultraviolet absorption and fluorescence detectors operating at the UV-Vis wavelength of 256 nm to exploit the highly unsaturated nature of humic substances and their characteristic feature of being fluorescent. However, some organic matter, including polysaccharides, cannot be detected using ultraviolet absorption or fluorescence, complicating the use of HPSEC to study the behavior of material such as highly hydrophilic macromolecular organic matter or low-molecular-weight organic acids. To address this difficulty, HPSEC-OCD, which uses an organic carbon detector (OCD), was developed.
OCDs are instruments that quantitate organic carbon content by measure the quantity CO2 released by the total oxidation of an organic material. In general, OCDs can measure all types of organic carbon with high accuracy. There are currently two types of OCDs: instruments based on dry oxidation, in which organic matter is oxidized by a high-temperature catalyst, and wet oxidation instruments, which oxidize organic matter using oxidizing agents and UV irradiation. Dry-oxidation methods offer the ability to measure the organic-matter content over a wide range of concentrations, ranging from a few hundred ppb to a few hundred ppm, whereas wet-oxidation methods offer high-sensitivity measurement of organic-matter content at low concentrations, ranging from a few ppb to a few ppm. An additional advantage of wet-oxidation methods is that they allow in-line measurements, facilitating connection to HPLC systems. 800 Series Total Organic Carbon Analyzer developed by Sievers is equipped with a turbo mode capable of making total organic carbon (TOC) measurements at 4-second intervals. A hybrid instrument consisting of a 800 Series Total Organic Carbon Analyzer connected to an HPLC system formed the basis of a novel technique for the analysis of humic substances and the results were published in Environmental Science and Technology in 20003). The ability to observe peaks that are not detected by ultraviolet absorption or fluorescence detectors has enabled the detection of hydrophilic behavior in macromolecules that could not have been observed previously.
Wet-oxidation OCD systems consist of several components connected end-to-end in a single-line configuration: a solution-sending pump, a degassing apparatus to remove inorganic carbon from the sample and eluent, a syringe pump to inject oxidizing agents and buffer liquids, a UV lamp to promote the oxidation reaction, and an Membrane Conductometric cell to measure CO2 gas quantities. In Japan, the M9e series of instruments (descendants of the original 800 Series Total Organic Carbon Analyzer) are available for purchase from the distributor Central Kagaku Corp. The procedural flow of a typical analysis is depicted schematically in the figure below.
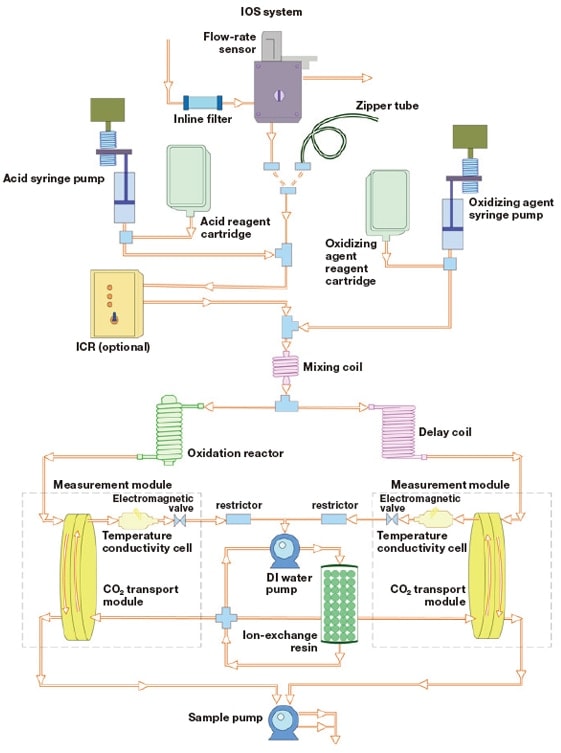
Fig. 2 Schematic procedural flow of an OCD measurement.
Flow rate
When interconnecting an OCD with an HPLC system, one parameter that requires particular attention is the rate of flow through the OCD. In the Sievers OCD, the solution-sending flow rate is fixed, and solutions separated via the HPLC columns must be supplied at this flow rate. Failure to supply a sufficient flow of solution may result in the injection of air into the OCD, potentially resulting in system malfunction (in the worst case). On the other hand, an excessive flow of solution may pressurize the solution-sending line, resulting in fluid leakage. The OCD solution-sending rate is also affected by factors such as deterioration of the tube pumps because one is increasing or decreasing the flow rate around a fixed value. Thus, in practice, a flow-rate-adjusting buffer is needed between the HPLC and OCD instruments. Our solution is to insert a three-way connector between the OCD and the HPLC; we have designed our system such that, when the HPLC flow rate exceeds that of the TOC, we divert some of the incoming flow in the form of waste liquid, whereas in the opposite scenario, we introduce a portion of the waste liquid into the OCD. In our experiments, we ensure that no waste liquid is introduced by adjusting the HPLC solution-sending rate to ensure that the HPLC flow rate is always slightly greater than that of the TOC. In practice, this requires some trial-and-error experimentation upon connecting the two systems.
Choice of columns
In choosing appropriate columns, there are three conditions that must be satisfied: (1) it must be possible to use inorganic eluents, (2) it must be possible to use the system at high flow rates of 1 mL/min or higher, and (3) the system must be able to separate organic matter with molecular weights ranging from a few hundred to a few million Da. In particular, when separating molecules of widely varying molecular weights at flow rates of 1 mL/min or above, the resin particles are relatively small, and one must take care to ensure adequate hold times within the columns. For this reason, a common practice—and one that has been adopted in prior studies—is to use an large column, 20 mm in diameter and 25 cm long, into which one injects Tosoh Corporation's Toyopearl® HW-50s resin. The exclusion limit of Toyopearl® HW-50s is 8 × 104 Da; in cases where one wishes to separate molecules of higher
molecular weight, one may instead use Toyopearl® HW-65s, with an exclusion limit of 5 × 106 Da. With a 20 mm × 25 cm column and a flow rate of 1 mL/min or higher, hold times within the column are long. As a result, analysis is time-consuming, requiring over 120 minutes to analyze a single sample, and the throughput is low (only 12 samples can be analyzed per day). For our system, we link two columns together in tandem, each 7.8 mm in diameter and 30 cm long, with Toyopearl® HW-65s injected into the upstream column and TSKgel® G3000SWXL injected into the other. This allows more rapid measurement of macromolecular organic matter and humic acid. Using this technique, it is possible to make measurements in less than half the time required conventionally (approximately 50 minutes), facilitating analysis of large numbers of samples. Thus, when selecting columns, it is possible to identify conditions enabling rapid analysis by carefully considering the precise range of molecular weights over which one needs good measurement resolution and by interconnecting multiple columns.
Sample injection volumes
The sample injection volume depends on the detector sensitivity. Eluent measurements via OCD have a sensitivity on the order of 20 ppb; no peaks will be detected for components with concentrations below this threshold. Using the large columns described above results in long hold times and thus dilution of the injected samples. Consequently, it is typically necessary to inject large sample volumes on the order of 1-5 mL. In contrast, the two-column technique that we have developed allows sample injection volumes to be reduced to as little as 1/3 the volume required for conventional methods, with typical sample injection volumes in our measurements ranging from 500 µL to 1 mL.
Oxidizing agents and pH buffer solutions for TOCs
The quantities of pH buffer solutions and oxidizing agents required for OCD analysis must be adjusted as appropriate for the sample in question; however, in cases where the water source and the organic-matter concentration being studied in a given experiment are fixed to some extent, there is almost no opportunity for adjustment after initial optimization. Typical values for water from rivers or lakes are 0.75 µL/min for the phosphoric acid buffer solution and 2.5 µL/min for the oxidizing agent.
Figure 3 shows the relationship between hold time and molecular weight for an HPSEC-OCD system equipped with our two-column system, in which the upstream column is injected with Toyopearl® HW-65s and the downstream column is injected with TSKgel® G3000SWXL.
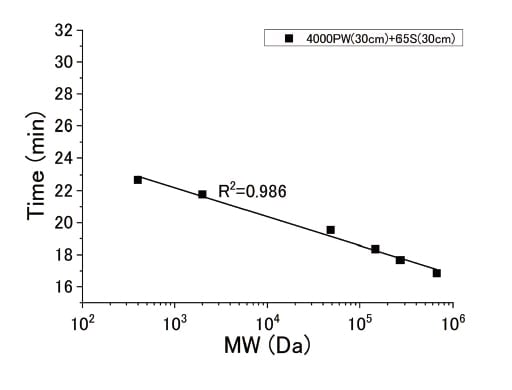
Fig. 3 Relationship between molecular weight and hold time
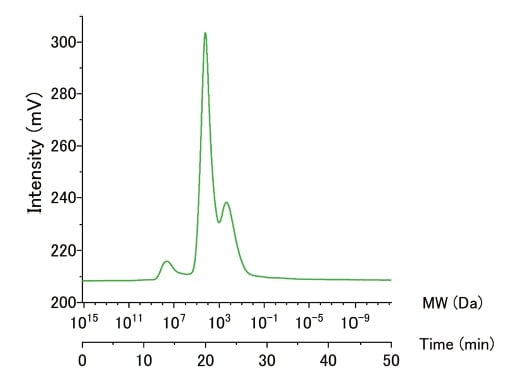
Fig. 4 Example of an HPSEC-OCD analyzer measurement
The HPLC used in this experiment was a Chromaster® high-speed liquid chromatograph from Hitachi.
This column system provides a linear relationship over a wide range of molecular weights, from a few hundred Da to a few hundred thousand Da, demonstrating the efficacy of our two-column configuration in separating a wide range of molecules. In particular, macromolecules with weights of 100,000 (105) Da or more are completely separated from molecules with weights of a few thousand Da, with no overlapping peaks, thereby allowing the quantitative determination of concentrations from the peak areas. Figure 4 shows the results of an analysis of the molecular-weight distribution in water samples taken from the Chitose River, which flows through the city of Ebetsu in Japan's Hokkaido region. Three prominent peaks were detected. The high second peak was also correctly detected by a UV detector but the lower first peak was not, suggesting that this peak may be due to polysaccharide-like components that do not absorb UV light. This example demonstrates that HPSEC-OCD allows the detection and quantitative measurement of macromolecule and polysaccharide components that cannot be detected by other detectors.
Returning to our earlier discussion of the origins of organic matter, it is well known that organic matter derived from animals is particularly rich in macromolecular components. Other sources of water often exhibiting similar richness include sewage-treatment facilities with high levels of biological activity, and nutrient-enriched lakes. In contrast, organic matter derived from plants typically decomposes over a longer period of time, yielding low concentrations of macromolecular components. Based on these observations, several research studies have reported that, by measuring the concentrations of macromolecular components detected using HPSEC-OCD, it may be possible to assess factors such as biological activity in water or the extent of man-made pollution5).
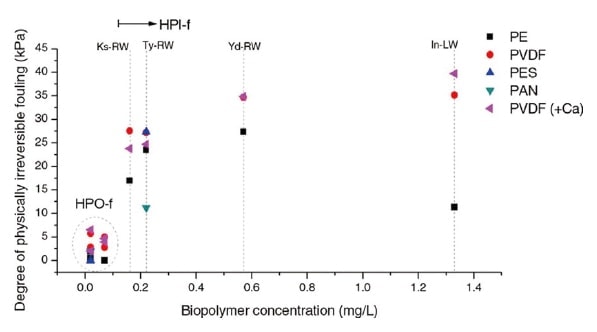
Fig. 5 Relationship between membrane fouling and the concentration of macromolecular organic matter5)
It is also well known that these macromolecular components clog the pores of filtration membranes in water-treatment and sewage-treatment plants, and a clear linear relationship has been demonstrated between the concentration of macromolecular components and the rapidity of membrane clogging. In view of this and other promising arenas for future applications of HPSEC-OCD analysis, which include food-producing industries and bioprocess management, we see that the technique is not limited to the analysis of river and lake water, but instead will find utility in a wide variety of fields.
Thus far, we have primarily emphasized the advantages of HPSEC-OCD, but the method also suffers from many drawbacks that must be rectified. The most critical problem is simply that of OCD downtime; the use of a syringe pump to inject oxidizing agents and pH buffer solutions (see Figure 2) requires that syringes be replenished with fluids at fixed time intervals, a process during which no data can be obtained. Of course, this poses no practical difficulty as long as system downtime does not coincide with peak measurements; however, when mandatory downtimes arise just as peaks are being detected, one must repeat the measurement of the sample in question from the beginning. This issue is being improved with each OCD model change. Also, when analyzing samples containing significant quantities of oils or other highly hydrophobic components, the tendency of these components to adhere to columns requires some pre-processing steps prior to analysis, as otherwise the lifetimes of the expensive columns may be greatly shortened.
Although in this paper we have thus far described the efficacy of the OCD as an HPSEC detector, in this section we discuss an alternative detector technology that we are currently developing: the HPSEC-three-dimensional excitation emission matrix (EEM), which pairs an HPSEC system with a fluorescence spectrophotometer. The EEM allows simple high-sensitivity analysis of humic substances and similar species and has been widely used to date for purposes such as water-quality analysis of rivers, lakes, and sea areas. In particular, recently developed techniques allow the separation of overlapping peaks using the multivariate analysis algorithm PARAFAC (parallel factor analysis)8). This advance has spurred the development of a wide range of applications, from process controls for water-treatment and sewage-treatment to the identification of pollution sources in aqueous environments. However, the use of PARAFAC to separate overlapping spectra requires the collection of large quantities of highly diverse samples, and in practice it is commonly difficult to separate components due to insufficient numbers of samples and/or sample populations of insufficient diversity. To address this difficulty, we are developing a method in which organic matter initially separated by size using HPSEC is subjected to EEM analysis, allowing application of accurate PARAFAC with fewer samples than are conventionally required.
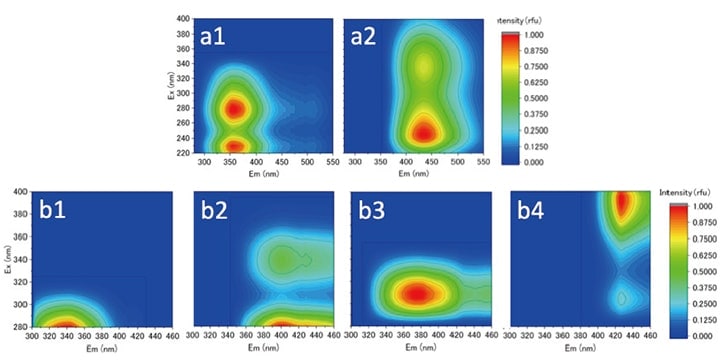
Fig. 6 Component compositions obtained by using PARAFAC to separate 4 types of samples. (a) Results of direct application of PARAFAC to obtained spectra. (b) Results of application of PARAFAC to EEM-measured spectra after LC separation and preparation of samples
More specifically, after separation by HPSEC, samples are gathered on microplates and a Hitachi F-7100 fluorescence spectrophotometer equipped with a microplate accessory is used to acquire three-dimensional EEM; for example, we successfully acquired 29 EEM spectra for samples. A direct application of EEM-PARAFAC to a collection of 4 types of samples, including water from sewage-treatment plants, succeeded only in separating the sample population into two components. However, an application of PARAFAC to the EEM-analyzed data following HPSEC separation was significantly more promising: it successfully separated four components, two more than were obtained via direct analysis. These results suggest that our new method may prove an extremely powerful technique for cases in which too few samples prevented successful application of PARAFAC in the past. Moreover, the possibility of conducting analyses using HPSEC and EEM instruments in tandem offers the promise of elucidating the behavior of organic matter in water with unprecedented speed and convenience. Currently we are developing an instrument that connects an HPSEC system to a fluorescence spectrophotometer equipped with a flow-cell accessory, and are focusing on determining the optimal conditions for measurement and analysis. We look forward to presenting further progress in the form of future reports in this magazine.
References
See more