In recent years, the field of chemical analysis has witnessed a number of difficult challenges—including an increasingly diverse variety of substances to be tested, a trend toward lower-concentration test samples, and evergrowing demands for higher-precision analyses—with the result that methods of instrumental analysis have become essential components for even the most routine, everyday measurements. In addition, the growing sophistication of analytical instruments has advanced the frontiers of measurement automation, while instrument operating manuals are now commonly made available for use in the process of certifying test facilities; thanks to these developments, instrument operators of all skill levels—including rank beginners—may now be expected to deliver reasonably
reliable results. However, instruments do not make measurements of substances themselves; rather, instruments measure relative numerical variations in physical quantities—such as currents or voltages—induced by various phenomena involving the substances in question. Thus, the concentration of a test sample cannot be determined simply by mounting the sample in an instrument. Nonetheless, in practice many quantities of interest—including environmental quality standards, water-quality standards, and product-quality standards—are characterized in terms of sample concentrations, and thus in many cases the key physical quantity we wish to determine is, ultimately, a concentration. Consequently, the experimental determination of concentrations requires a preliminary calibration step, in which a reference material is used to construct calibration curves relating values read from instruments to the actual concentrations of test samples. For that reason, the accuracy of calibration curves—and thus the accuracy of all measurements—is heavily affected by the quality of reference materials.
In this article we first present an overview of reference materials—and their essential role in ensuring reliable results for chemical analysis—then discuss a number of guidelines to keep in mind when working with standard solutions.
The phrase reference material is a blanket term that may refer to any of a large number of substances, chosen as appropriate for the objective at hand.
The reference materials used in chemical analysis may be broadly classified into two categories: pure reference materials and matrix reference materials. Examples of the former include high-purity substances and standard gases or standard solutions prepared by diluting high-purity substances. Common varieties of standard solutions include pH standard solutions and inorganic standard solutions—prepared by dissolving high-purity reagents in water or acid—as well as organic standard solutions, prepared by dissolving reagents in organic solvents.
The term “matrix reference material” is a catch-all phrase encompassing all reference materials other than pure reference materials; in general these substances involve components of fixed concentration or composition embedded in a matrix. Examples include metallic reference materials such as steel products, inorganic reference materials such as ceramics, environmental reference materials such as seawater or soil samples, macromolecule reference materials such as synthetic polymers, and clinical reference materials such as blood serum.
The primary uses of reference materials include calibrating analytical instruments and measurement equipment, assigning values to other substances, validating laboratory instruments and experimental procedures, and verifying technical details such as internal and external quality control; in general, each such task will entail a different choice of reference material, selected as appropriate for the objectives at hand. In particular, the pure reference materials used to calibrate instruments are often used to determine the characteristics of matrix reference materials—again testifying to the essential role of reference materials in determining the results of experiments.
In ISO guide 30:2015 (JIS Q 0030:2019), “Reference materials—Selected terms and definitions,” the term “reference material'' (RM) is defined as follows: “material, sufficiently homogeneous and stable with respect to one or more specified properties, which has been established to be fit for its intended use in a measurement process.’’
The term “Certified reference material'' (CRM), used to refer to reference materials that have been vetted particularly rigorously, is also defined by this document: “reference material (RM) characterized by a metrologically valid procedure for one or more specified properties, accompanied by an RM certificate that provides the value of the specified property, its associated uncertainty, and a statement of metrological traceability.’’
Thus, CRMs have established metrological traceability and come with uncertainty indicating the accuracy with which their properties have been characterized. ISO/IEC 17025:2017 (JIS Q 17025:2018), “General requirements for the competence of testing and calibration laboratories,” describes how accepted values for the properties of a CRM— provided by a competent manufacturer with metrological traceability to declared quantities in the International System of Units (SI units)—may be used to furnish one definitive proof that the results of measurements are traceable in SI units. Thus, we see that CRMs are required to yield measurement results of high reliability.
As noted above, in practice there are many substances known as “reference materials”—including both reference materials guaranteed by producers of reagents, and matrix reference materials supplied by academic societies used at experimental facilities or for assessing the skills of analytical personnel—and the metrological traceability of many of these is unclear. In contrast, metrological traceability is clearly established for the standard gases and standard solutions provided by the metrological traceability framework of the Japan Calibration Service System (JCSS)— discussed below—as well as for CRMs provided by the National Metrology Institute of Japan / Japan’s National Institute of Advanced Industrial Science and Technology (NMIJ/AIST). In practice, one must make choices appropriate for the task at hand, remaining cognizant that different reference materials offer different levels of reliability.
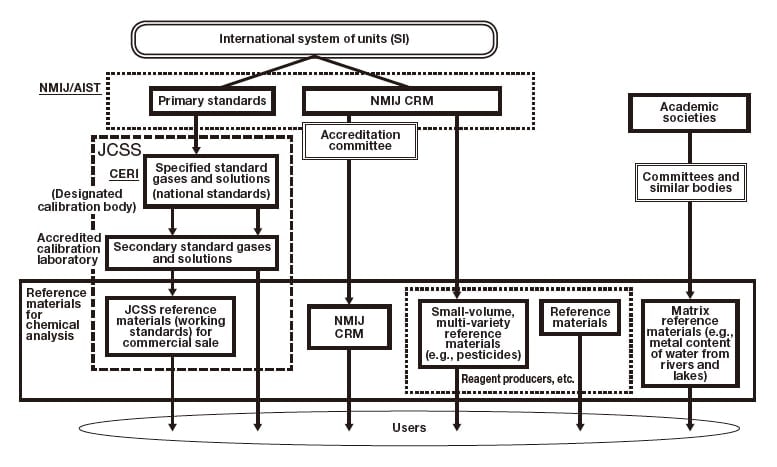
Fig. 1 The hierarchical supply chain for reference materials in Japan.
NMIJ/AIST: National Metrology Institute of Japan / National Institute of Advanced Industrial Science and Technology
NMIJ CRM: Certified reference material provided by the NMIJ
CERI: Chemicals Evaluation and Research Institute, Japan
JCSS: Japan Calibration Service System
JCSS reference materials for practical applications: Commercially-available reference materials provided through the metrological traceability framework
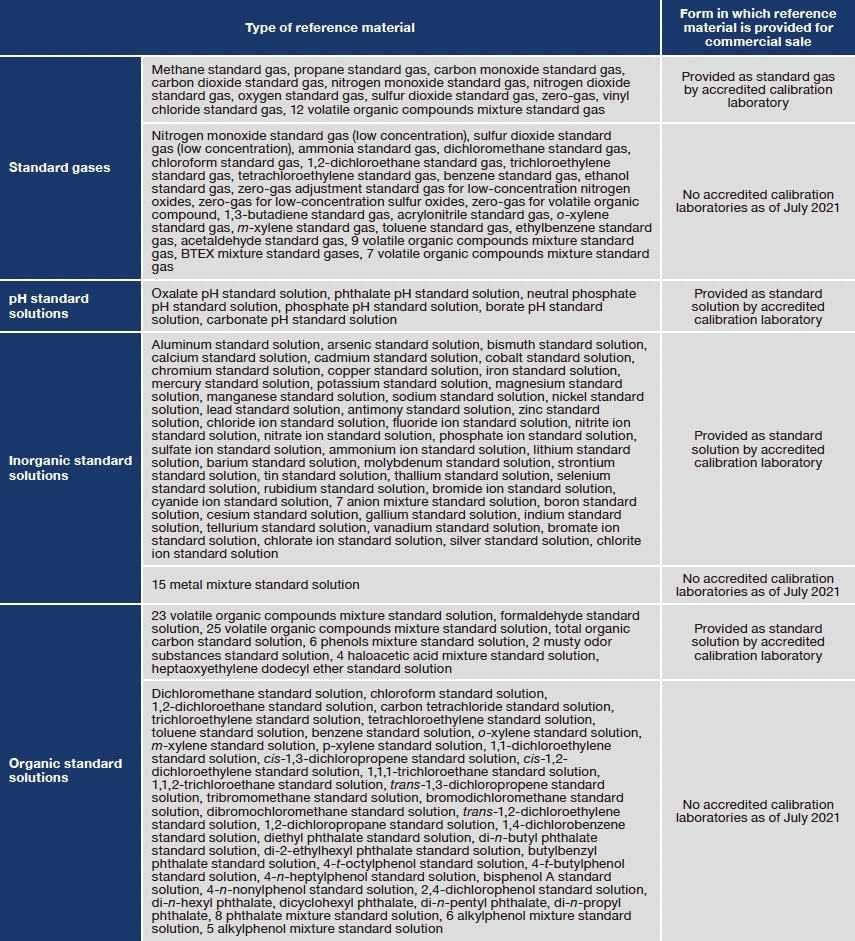
We will use the term JCSS reference materials to refer to reference materials provided by the Japan Calibration Service System (JCSS), a metrological traceability framework created in 1993 by legislation amending Japanese Measurement Act. The JCSS provides metrological standards for length, mass, time, and 22 other physical quantities, one of which is concentration (of reference materials). Supplies of JCSS reference materials are made possible by the following hierarchical framework. First, designated calibration body [Chemicals Evaluation and Research Institutes, Japan (CERI)] is designated by Japan’s Ministry of Economy, Trade, and Industry to preparation and maintain specified standard gases and solutions —corresponding to national standards—and to calibrate (and assign values to) secondary standard gases and solutions prepared by accredited calibration laboratories. The accredited calibration laboratories then use their secondary standard gases and solutions to calibrate (and assign values to) working standard gases and solutions for practical applications, which are accompanied by calibration certificates—stamped with the official JCSS logo for authentication—and made available for commercial sale. Because the concentrations of Specified standard gases and solutions are traceable to the international (SI) system of units through CRMs from the National Metrology Institute of Japan (NMIJ), the calibration hierarchy ensures that the concentrations of JCSS reference materials are traceable to SI units as well. Thus, JCSS reference materials have clearly traceable metrological pedigrees and serve as powerful tools for ensuring the trustworthiness of measured data. As of July 2021, a total of 135 varieties of designated specified standard gases and solutions —including 34 standard gases and 101 standard solutions—have been established.
Table 1: Specified standard gases and solution
When handling reference materials, it is essential to read and obey all instructions—listed on material certificates or other documents—regarding the storage and usage of the material (such instructions may include preprocessing conditions such as drying protocols, or guidelines such as minimum sample volumes).
In general, most commercially available standard solutions are provided in concentrations of 1000 mg/L or 100 mg/L. Documents in the series JIS K 0010 - JIS K 0038 (deprecated in 2007) are standards for inorganic standard solutions— such as pH standard solutions, copper standard solutions, or nitrate ion standard solutions—and list the following precautions to observe when using these substances:
The reason for requiring solutions to remain unfrozen is that the freezing process may induce clumping or other processes that degrade the uniformity of material compositions. Storage vessels with screw-on caps often have lids whose material composition differs from that of the vessel body; in such cases, the difference in shrinkage factor of the two materials may cause gaps to form between body and lid. This, in turn, may result in vaporization or other processes that might affect the concentration of solutions. Another concern is the possibility that solutions in vessels may develop concentration inhomogeneities due to factors such as variations in the density of solution components or adsorption of solution on the inner walls of vessels; to avoid such effects, containers of reference materials should be thoroughly agitated before they are opened. Needless to say, one must also observe basic precautions: containers should not be opened until immediately before their contents are used, and substances should not be used after their shelf life has expired.
At present, organic standard solutions such as the 25 volatile organic compounds mixture standard solution provided by the JCSS are stored at refrigerated temperatures. Also, organic standard solutions and other substances kept in ampoules should be used immediately upon opening the ampoule; any leftover substances remaining after completion of experiments should be discarded, and not saved for reuse.
The general guidelines we have outlined here are only representative examples of usage and storage conditions; when working with actual substances, the most important step is to read and understand the full text of the material descriptions included in material certificates or printed on labels affixed to containers.
In instrumental analysis, one typically constructs calibration curves by diluting a standard solution (stock solution)—which will generally be of a standard concentration such as 1000 mg/L—to prepare standard solutions for calibration curves. A variety of precautions must be observed to prepare standard solutions capable of yielding calibration curves for precise concentrations.
The standard solution used as the stock solution may be a single-component solution or a mixture solution, but in either case one generally starts with a high-concentration standard solution, which is then diluted by adding water or acidic solutions.
The quality of the water or acid used as a diluent in this process is a key factor that must not be overlooked if one hopes to prepare standard solutions for calibration curves with precise concentrations. If the diluent contains impurities with compositions matching any component of any substance to be measured, accuracy will be lost when attempting to determine the concentration of calibration-curve solutions. The purity of water or acid diluents is thus of critical importance, and—depending on the goals of the measurement in question—may require special quality controls.
A standard for water used in chemical analysis is JIS K 0557, “Water used for industrial water and wastewater analysis,” which establishes 4 varieties by type and quality; however, in practical use, quality requirements vary depending on the objective in question, necessitating blank test to be filled in as appropriate for the task at hand. Also, when water is set aside and stored for long periods of time, contamination and various other problems tend to arise, so it is desirable to use water as soon as possible after its production.
For substances such as metals, standard solutions for calibration curves are typically prepared with nitric acid or other acids; the quality of the acids used in such cases must be evaluated as carefully as the quality of the water used to prepare aqueous solutions.
Nitric acid is discussed by (at least) two distinct JIS standards, JIS K 8541 “Nitric acid (reagent)” and JIS K 9901 “Highly purified nitric acid”; these two standards target products have different quality levels, and correspondingly impose impurity-content regulations of differing stringency. There also exist products produced to the specifications of reagent manufacturers themselves; for example, certain acids—intended for use in high-sensitivity analysis—are subject to stringent restrictions on the concentrations of various impurities, which are controlled at levels ranging from ppb (by mass) to ppt (by mass). The information of impurity concentrations, etc., may be obtained not only by looking up specifications on the websites of reagent producers, but also by acquiring certification of analysis for individual production lots.
Organic standard solutions are prepared using organic solvents such as methanol and hexane; as we noted previously for acids, solvents such as these are commercially available in a variety of forms—from various reagent producers, for various purposes, produced to various specifications—and one must carefully evaluate the available options, considering impurity content and other data, to select the optimal product for a given application.
For dilution, the volumetric method, typically using glass measurement vessels such as volumetric pipettes and volumetric flasks, is widely used. Information on the accuracy of volumetric glassware may be obtained from manufacturers or from sources such as JIS R 3505, “Volumetric Glassware.” The marked line on volumetric glassware are calibrated in a 20°C environment; thus, when diluting standard solutions, one must consider temperature of not only the volumetric glassware but also both the standard solution (the stock solution to be diluted) and of the diluent. In particular, when preparing acidic solutions for use as diluents, the addition of acid to water will result in a release of heat; for this reason, it is best to prepare acid solutions in advance, allowing enough time for the temperature of the solution to stabilize before it is used. Other miniature volumetric tools such as piston pipettes, which are easier to use than volumetric pipettes, have recently become quite popular; however, the fact that these tools handle only minuscule quantities of substances require special care to ensure that volumes of liquid samples are characterized with sufficient accuracy.
Volumetric glassware, as well as piston pipettes, may be calibrated as necessary before use. Calibration methods for volumetric glassware are discussed in JIS K 0050, “General rules for chemical analysis,” while calibration methods for micropipettes are specified in JIS K 0970, “Piston pipettes.”
Of course, glassware-based volumetric method is not the only approach to dilution; one common alternative is the method known as gravimetric mixing, in which precision balance are used to measure portions of solutes and solvents with well-characterized masses. A typical workflow for dilution by gravimetric method is as follows:
Whereas the accuracy of volumetric method is affected by the accuracy of the volumetric glassware and other factors, the accuracy of gravimetric mixing is determined solely by the accuracy of the mass balance and the density meter, and thus gravimetric method generally achieves greater accuracy than volumetric method. Nonetheless, the performance of volumetric mixing is usually perfectly adequate for typical tasks in instrumental analysis. In dilution by gravimetric method, quantities of solutes and solvents are characterized by mass, so relative proportions take the form of dimensionless ratios, i.e., quantities with units of g/g; converting these values into quantities with dimensions of concentration (i.e., g/L) requires measuring densities. However, these measurements do not necessarily require the use of volumetric glassware, and may be implemented by a wide variety of tools—a fact that accentuates the broad applicability and general-purpose nature of gravimetric methods. For example, in cases involving highly volatile organic substances, the use of volumetric flasks to prepare standard solutions may incur solvent losses due to evaporation from regions near the interface between the flask and the lid, potentially degrading the accuracy of concentration measurements. In contrast, diluting standard solutions by gravimetric method allows the use of tightly sealed containers and eliminates uncertainties associated with gradation markings on volumetric glassware, expanding the range of possible dilution procedures.
Conventional analytical methods such as light-absorption spectroscopy and atomic absorption spectroscopy generally make use of single-component standard solutions. More recently, however, there has been surge of interest in techniques—such as inductively coupled plasma atomic emission spectrometry (ICP-AES) and inductively coupled plasma mass spectrometry (ICP-MS)—allowing multiple elements to be measured simultaneously. Calibration curves for analyses of this sort are typically constructed using multi-component standard solutions. In principle, one might attempt to prepare such solutions simply by combining two or more single-component solutions, in that case, it is necessary to pay attention to the formation of insoluble substances.
For example, attempting to mix a chromium standard solution with a lead standard solution results in the production of lead chromate, a substance that remains insoluble in solvents. Having said that, we note that lead chromate is produced in cases where chromium is present in a hexavalent configuration, and that the progress of lead chromate formation varies depending on factors such as the concentration of the solutions at the moment of mixing and the time elapsed since that moment. Thus, by diluting the standard solutions before their use it may be possible to avoid the production of insoluble substances.
In some cases, depending on the kind of solvent for the standard solution, insoluble substances may be produced. Most standard solutions are nitric-acid solutions, but may be provided in the form of nitric-acid, hydrochloric-acid, or sulfuric-acid solutions due to problems involving concentration stability and other issues depending on component varieties. Thus, when mixing standard solutions of different solvent, reactions with components of measurement samples may give rise to insoluble products. For example, arsenic standard solutions are provided as hydrochloricacid solutions; upon mixing with silver standard solutions, chlorine reacts with silver to yield insoluble silver chloride. Thus, when preparing standard solutions one may not simply mix together single-component solutions arbitrarily, but must consider the components of solutions listed on their labels or elsewhere and assess the possibility of reactions between mixed solutions and the solubility of any precipitates that may be formed as reaction products.
When a standard solution is retrieved from long-term storage and used to make measurements, no effort is made to check for, or correct, discrepancies between the actual concentration of the stored solution and the nominal concentration at which it was prepared or labeled (before storage). Thus, it is important to store solutions under conditions that prevent their concentration from varying over time; indeed, concentration measurements based on calibration curves created using incorrectly-concentrated standard solutions are clearly incapable of producing reliable results. This requires accurately assessing the stability of stored solution concentrations. For JCSS reference materials, storage stability tests are conducted at the development stage, and optimal storage conditions are determined through comparative assessments of storage containers, storage temperatures, solvent of dilution, and other relevant considerations.
Factors that may affect the stability of stored solution concentrations include the following:
These factors interact in complicated ways with sample components, environmental conditions, and other variables, and general trends are difficult to identify. The best strategy is to assess the stability of stored solution concentrations on the basis of usage methods, storage methods, environmental conditions, and other relevant factors.
The stability of a storage protocol is generally tested by preparing and storing a sample, using standard solutions for calibration curves to make independent easurements of sample concentration at the beginning of the storage interval and again after a given interval has elapsed, and comparing the initial and subsequent measurements to characterize any change in concentration.
As one illustration of the stability of stored solutions, Figure 2 shows temporal variations in the concentration of a standard solution of cyanide ions. In storage stability tests for this solution, we identified four factors thought likely to influence storage outcomes: the concentration of the standard solution, the concentration of sodium hydroxide, the type of container used to store the solution, and the storage temperature. For each of these variables, we considered 2 or 3 distinct values or selections, yielding an ensemble of storage parameter configurations. For each configuration in this ensemble we prepared a standard solution of cyanide ions, stored it under the conditions of the configuration, and measured temporal variations in concentration over a 6-month interval. Finally, we conducted a statistical analysis of our results to quantify the influence of each parameter on storage stability, and then used our findings to identify the optimal settings for all parameters. Some of our results are displayed in Figure 2. For solutions stored at 5°C, concentrations varied by less than 5% over 6 months. On the other hand, for solutions stored at 40°C the variation in concentration was around 80% over 6 months. These results indicate that storage temperature is the most important factor governing the stability of stored solution concentrations.
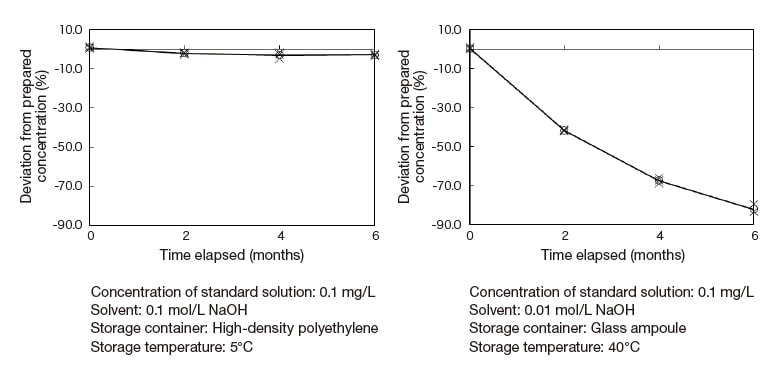
Fig. 2 Stability of cyanide-ion solution concentration
Standard solutions for producing calibration curves are usually prepared when they are to be used, but in some cases may need to be temporarily stored. If the solution was prepared in a volumetric flask or similar container, it may seem convenient simply to store it in that container, but for multiple reasons this turns out to be impractical. One difficulty is the possibility that some of the solvent may evaporate from the stem region of the flask, changing the concentration of solution away from its known value. A different risk is posed by the possibility of elements being eluted from the volumetric flask itself, creating a separate set of problems. Thus, the best policy is to store solutions in separate storage containers that can be tightly sealed.
In general, storage containers may be made from any number of materials, including glass, polyethylene, and polypropylene. In addition to its nominal material composition, each vessel typically contains other substances such as impurities in component materials or manufacturing additives, which may elute into standard solutions stored in the container. In particular, the elution of measurement sample components from containers containing standard solutions for producing calibration curves may affect the accuracy of the curves and must be carefully monitored.
Also, the high-density polyethylene containers commonly used as storage containers for standard solutions may allow some evaporation of solvents from inner container walls or other sites. This may be due to aqueous components of standard solutions passing through container walls as water vapor or in other forms. As we see from the diminishing concentration of cyanide-ion standard solutions in Figure 2, solution concentrations may change over time even in the absence of decomposition reactions or other phenomena directly tied to concentration, and must therefore be carefully monitored.
Reference materials are essential for the measurements that scientists and engineers perform on a daily basis. Reference materials exist in many distinct forms—and offer widely varying levels of reliability—and must be chosen judiciously in accordance with the goals of the analysis in question. Even the most dependable reference materials can be rendered unreliable by improper handling or incorrect use. Ultimately, measurements can yield trustworthy results only when the experimentalist devotes proper attention to choosing and using reference materials commensurate with the goals, samples, and measured concentrations of each analysis.
References
See more