3. The semiconductor material silicon
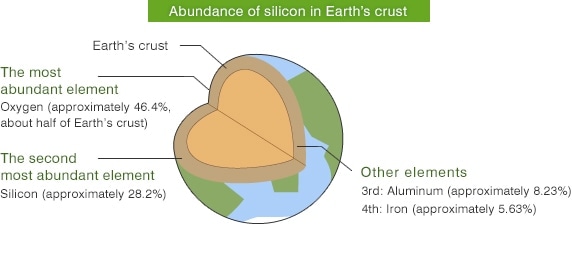
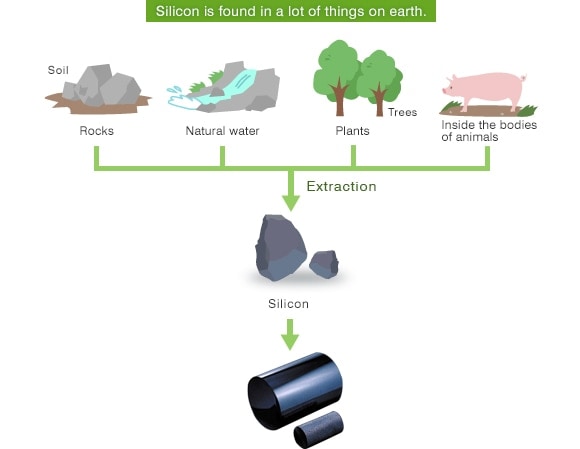
Semiconductors are widely used in familiar electric appliances such as personal computers, televisions, smartphones, digital cameras, IC cards, etc. The material most frequently used in semiconductors is Silicon (chemical symbol = Si). Silicon is the second most abundant element on earth after Oxygen. Most Silicon is found in soil and rock, but Silicon is also contained in natural water, trees and plants.
In nature, however, Silicon is found in the form of compounds with Oxygen, Aluminum and Magnesium. As a result, the Silicon element must be extracted from the compound and purified. Silicon used in a semiconductors such as an integrated circuits (IC) requires a single crystal structure of ultra-high-purity “99.999999999%” (the so-called “eleven nines”) and is refined using various processes after extraction.
A single crystal structure is a structure whereby atoms are arranged in an orderly fashion in three dimensions, and the basic unit of the arrangement is called the crystal lattice. A single crystal is a crystal lattice in an orderly, continuous arrangement. A Silicon crystal lattice has a diamond cubic crystal structure in a repeating pattern of eight atoms. Each Silicon atom is combined with four neighboring silicon atoms by four bonds. Silicon, a very common element, is used as the raw material of semiconductors because of its stable structure.
Purification of Silicon consumes large amounts of power. In Japan, refined Silicon (ingot) with purity of at least 98% is imported from Australia, China and Brazil, where electricity is relatively inexpensive.