Principle of Thermal Analysis
Definition of Thermal Analysis
A group of techniques in which a property of the sample is monitored against time or temperature while the temperature of the sample, in a specified atmosphere, is programmed.
It is stated in gFor Better Thermal Analysis and Calorimetryh, 3rd Edition, published in 1991 by ICTAC (International Confederation for Thermal Analysis and Calorimetry).
This defines Thermal Analysis includes all the methods of measuring the sample properties while the sample temperature is program-controlled.
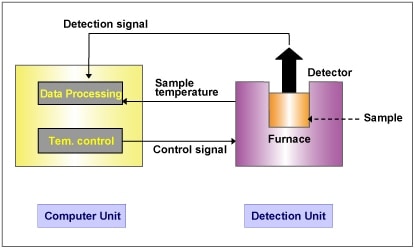
Figure 1 shows the configuration of Thermal Analysis instrument.
Thermal Analysis instrument includes the followings:
- Detection Unit: Furnace, sample and reference holder, and sensor, heat and cool the sample in the furnace, and detects the sample temperature and property.
- Temperature Control Unit: Controls the furnace temperature.
- Data Recording Unit: Records the signals of sensor and sample temperature, and analyzes them.
Temperature control, data recording and analysis are all computer-controlled. The combination of the furnace and sensor enables the various types of the measurement techniques. This computer can be connected to the several instruments which has the other types of measurement techniques, enables the simultaneous measurement and analysis.
| Abbrev. | TA Measurement Technique | Property | Unit |
|---|---|---|---|
| DTA | Differential Thermal Analysis | Difference temperature | °C or µV* |
| DSC | Differential Scanning Calorimetry | Enthalpy | W = J/sec |
| TG | Thermogravimerty | Mass | gram |
| TMA | Thermomechanical Analysis | Deformation | meter |
| DMA | Dynamic Mechanical Analysis |
Elasticity | Pa = N/m2 |
* Thermocouple electromotive force.
| Phenomenon/Property | Technique |
|||
|---|---|---|---|---|
| DSC | TG | TMA | DMA | |
| Melting | ⚪ | — | Δ | Δ |
| Glass transition | ⚪ | — | ⚪ | ⚪ |
| Crystallization | ⚪ | — | Δ | ⚪ |
| Reaction (Curing / Polymerization) | ⚪ | Δ | ⚪ | ⚪ |
| Sublimation / Evaporation / Dehydration | Δ | ⚪ | — | — |
| Thermal decomposition | Δ | ⚪ | — | — |
| Thermal expansion / Thermal shrinkage | — | — | ⚪ | — |
| Thermal history | ⚪ | — | ⚪ | ⚪ |
| Specific heat capacity | ⚪ | — | — | — |
⚪:Measurement Object
Δ:Some compatible
—:Not measured
The main application areas of DSC concern the determination of the melting, the glass transition, the crystallization, the chemical reactions, the thermal history, and the specific heat capacity. Though the sublimation, the evaporation, and the thermal decomposition can also be measured, this technique is not usually used because of the mass change by the decomposition and the corrosion of the sensor by the decomposed gas.
TG measures the mass changes by the evaporation, the dehydration, the decomposition, and the oxidation reaction. By the simultaneous measurement by TG connected with DTA, the exothermic and the endothermic phenomena can be measured from DTA.
TMA measures the phenomena caused by deformation including the thermal expansion, the thermal shrinkage, the glass transition, and the curing reaction. Though the melting and @the crystallization can measured, there is case of the adhesion of the sample to the probe by the melting.
DMA measures the changes in the molecular motion and the molecular structures such as the relaxation, the crystallization, and the curing reaction. It also measures the melting at the early stage until the melt flow occurs.
