Amino Acid Analysis Basic Course
1. Principle and basics of amino acid analysis
Generally, organic compounds that contain -COOH (carboxyl group) and -NH 2 (amino group) in the molecular structure are called amino acids. It is said that there are approx. 500 types of naturally occurring amino acids, including the approx. 20 types of amino acids that make up proteins.
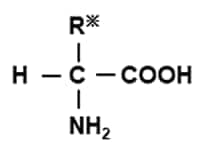
Individual organic groups are bonded to R.
Characteristics of each amino acid differ depending on the nature of R.
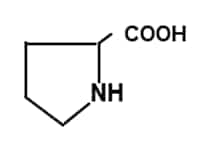
Proline has a ring structure (imino group). it shows specific optical properties different from other amino acids.
Amino acids are represented by three-letter code almost unified worldwide.
Table 1 Three-letter codes of amino acids
| Asp | Aspartic acid | Val | Valine |
|---|---|---|---|
| Thr | Threonine | Met | Methionine |
| Ser | Serine | Ile | Isoleucine |
| Asn | Aspargine | Leu | Leucine |
| Glu | Glutamic acid | Tyr | Tyrosine |
| Gln | Glutamine | Phe | Phenylalanine |
| Pro | Proline | Lys | Lysine |
| Gly | Glycine | His | Histidine |
| Ala | Alanine | Trp | Tryptophan |
| Cys | Cystine | Arg | Arginine |
In the field of amino acid analysis, Asparagine (Asn) and Glutamine (Gln), may be written as AspNH2 and GluNH2. Similarly, when described as Cys, it may refer to Cysteine.
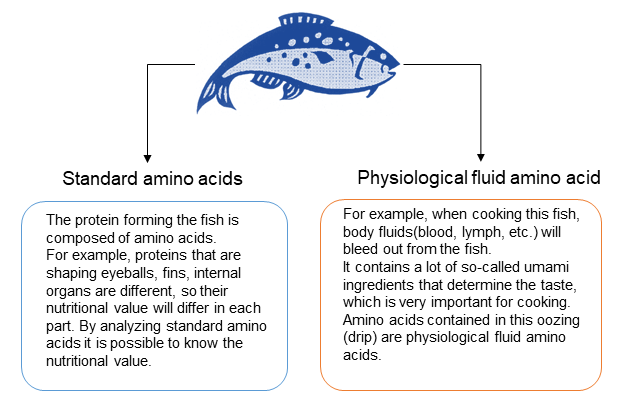
1) Standard amino acids (Protein-constituting amino acids, Proteinogenic amino acids)
The proteins that make up all animals are composed of amino acids. Amino acids that make up the protein are called standard amino acids (Protein-constituting amino acids, Proteinogenic amino acid). It is said that there are 20 standard amino acids.
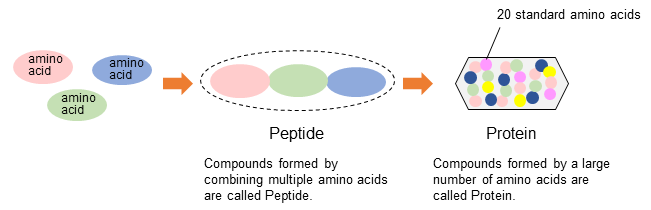
2) Physiological fluid amino acids (free amino acids)
The basic amino acids are 20 standard amino acids mentioned above, but there are many other amino acids that exist alone in living organisms and in nature. These amino acids, amino acid metabolites, and precursors are called physiological fluid amino acids (free amino acids). Generally, we analyze about 40 physiological fluid amino acids.
2. Amino acid analysis methods
Most amino acids are highly hydrophilic, have low UV absorption, and are not fluorescent, making them difficult to separate and detect. Therefore, they must be derivatized (labeled) before detection. Derivatization transforms them into compounds with chemical characteristics such as UV absorption and fluorescence, making them possible to separate and detect.
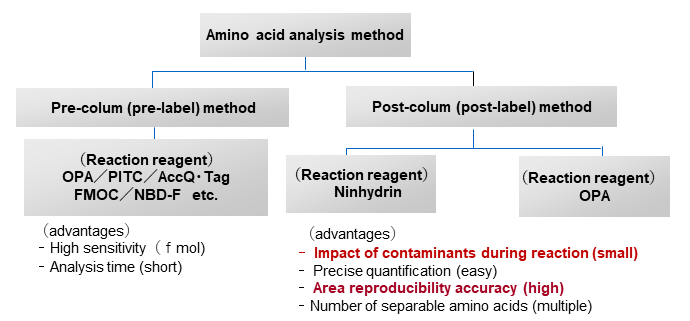
There are two main types of amino acid derivatization methods: post-column and pre-column derivatization, and the derivatization reagents (labeling reagents) used differ.
3. Pre-column and post-column derivatization methods
The two derivatization methods have different features. Pre-column derivatization, in which amino acid components are derivatized before column separation, requires a wide variety of reaction reagents and a relatively short analysis time, but has low reproducibility and is affected by impurities, making it more suitable for qualitative rather than quantitative analysis. On the other hand, post-column derivatization, in which derivatization is performed after column separation, is less affected by impurities and has high reproducibility, making it possible to precisely quantitate many amino acids, including free amino acids.
We will summarize pre-column and post-column derivatization methods.
Table 2 Amino acid derivatization methods and features
| Items | Amino Acid Analysis method | ||
|---|---|---|---|
| Derivatization method | Pre-column derivatization method | Post-column derivatization method | |
| Reaction Reagents | OPA/PITC/NBD-F FMOC/AccQ・Tag etc. |
OPA | Ninhydrin |
| Features |
- Reagent types (many)
- Analysis time (short) - Effect of impurities (high) - Separable amino acids (few)* - Reproducibility (low)* *Post-column derivatization method ratio |
- High sensitivity - Does not react with secondary amines - Reagents and reaction products deteriorate quickly |
- Quantitative accuracy (high) - Effect of impurities (low) - Separable amino acids (many) - Analysis time (long) |
| Instruments | - HPLC system - UHPLC system - LC-MS |
- HPLC system | - Amino Acid Analyzer (dedicated instrument) - HPLC system |
4. Method for analyzing amino acids
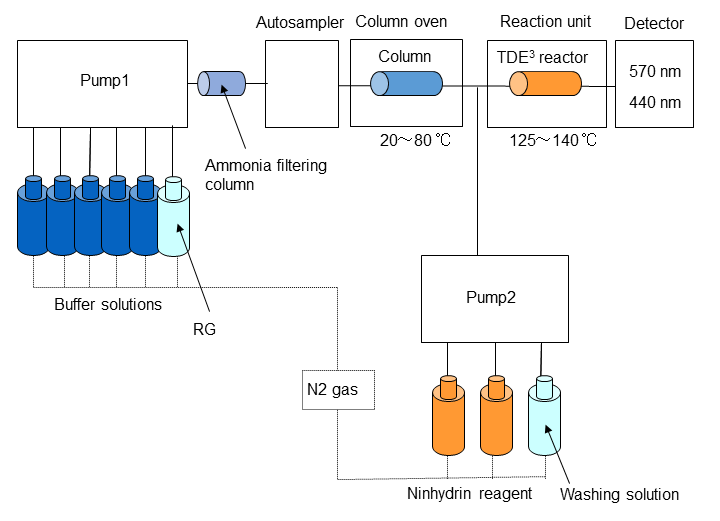
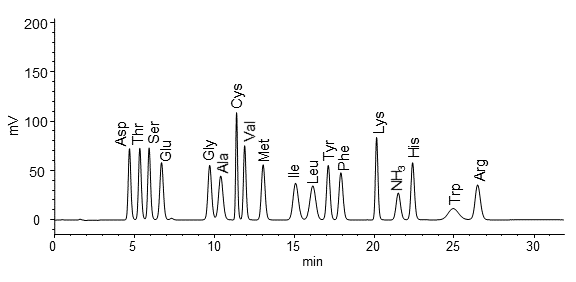
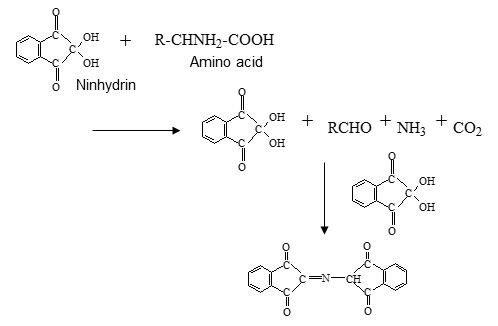
Hitachi‘s high-speed amino acid analyzer uses a cation exchange column for pH gradient separation and post-column derivatization using ninhydrin reagent, resulting in excellent quantitative accuracy and reproducibility. The reagents are also highly stable, allowing for simultaneous analysis of over 40 amino acids.
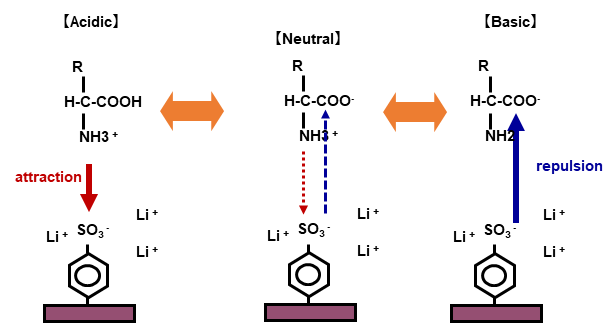
Reference:Cation exchange column and separation of amino acids
- Amino acids are charged to + in acidic solution, and electric attraction is generated to cation exchange resin.
- In neutral solution, amino acids have both + and - charges in the molecule, weakening their attraction to cation exchange resin.
-
In basic solution, amino acids are charged to -, so that they repel and pass through cation exchange resin.
(Utilizing this chemical property, we use sodium hydroxide solution (basic solution) for washing out remaining substances in the column after analysis. It is called regeneration (RG) process.) - In amino acid analysis, separation is carried out by changing pH of the eluent from acidic to basic.
The cation exchange column has a high chemical strength such as being able to wash with basic solutions, but the physical strength is not so high.
Column pressure must be monitored carefully, as sudden pressure fluctuations may cause deterioration.
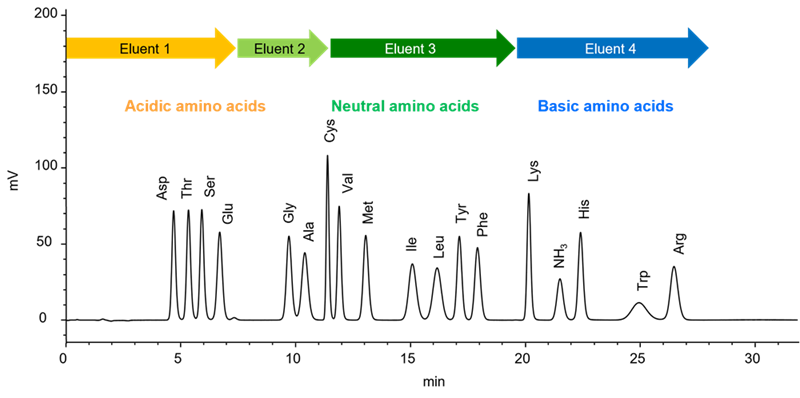
Table 3 Necessary elements for amino acid analysis buffer
| Condition | Chemicals |
|---|---|
| pH | Citric acid |
| Ionic strength | NaCl, LiCl |
| buffer capacity | Sodium citrate, Lithiun citrate |
Table 4 Other components of amino acid analysis buffer and purpose of addition
| Reagents | Porpose |
|---|---|
| Ethanol | separation of Thr-Ser |
| Benzyl alcohol | separation of Trp |
| β-thiodiglycol | anti-oxidation of sulfur-containing amino acids |
| Brij-35 | Pump pressure reduction |
| Caprylic acid | Anti-corruption |
In case of using the Hitach High-Speed Amino Acid Analyzer,the buffer solutions and reaction reagent are provided as MCI BUFFER™ (PH Kit, PF Kit) and Ninhydrin Coloring Solution Kit for HITACHI from FUJIFILM Wako Pure Chemical Corporation. The buffer solutions can be prepared according to “Buffers preparation” section of the instruction manual (main unit). At that time, pH described in the instruction manual is a reference value and adjustment of pH is not necessary. Please use special grade reagents or grade for amino acid analysis reagents.

