Accurate measurement of spectra for Fluorescence Spectrophotometer
A decrease in irradiated light is measured by absorption spectroscopy, while luminescence is observed by the fluorescence method; therefore, the wavelength differs between irradiated and measured light.
The fluorescence method has the advantage of being more sensitive (measurable at a low concentration) and more informative (acquisition of excitation and fluorescence spectra), as compared with the absorption spectroscopy.
However, wavelength characteristics specific to optical components of a fluorophotometer, such as a lamp, may cause differences among devices, and multiple lights such as secondary light, due to the effects of light scattering, may disturb measurement. Here, this is to introduce you a method for correcting (reducing) the differences among devices and a method for eliminating light scattering.

Effect to use Filter set
In surface photometry, multiple lights such as secondary light, due to the effects of light scattering, disturb measurement.
A cut filter is effective for eliminating multiple lights.
We utilize the Stoke's principle that fluorescence can be measured at a longer wavelength, as compared with excitation light.
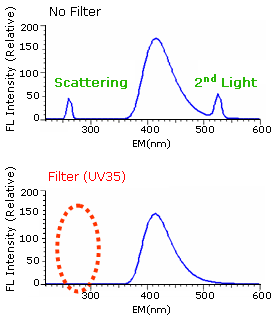
We'll introduce the effects of the cut filter using data obtained for sodium salicylate (powder). Use of a filter (UV35) cuts light scattering, led to a detector, to control the generation of secondary light.

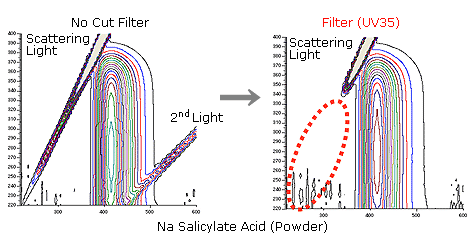
This is data extracted from three-dimensional data. From two dimensional data, the 2nd light is eliminating.
Effect to use Spectra Correction Kit
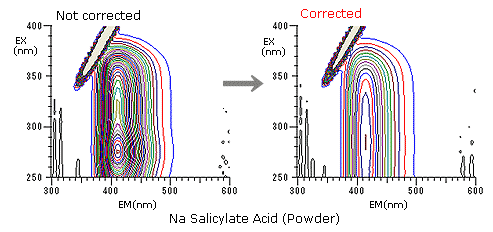
This is to introduce you the spectrum correction to get the accurate spectrum. Each lamp or detector of a fluorophotometer has its own wavelength characteristics.
Since the measured spectrum data reflects such characteristics, correction is needed to obtain a true spectrum. Here, we show the effects of a spectrum correction kit that generally includes a rhodamine B concentrated solution and a substandard light source.
These are the effects after spectrum correction using data obtained for sodium salicylate (powder). Three-dimensional data varied with the presence or absence of correction, demonstrating the effects of wavelength characteristics.

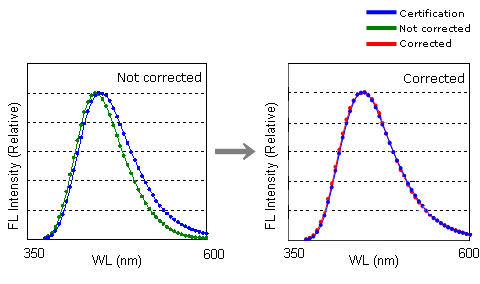
This is a comparison between literature data and measurement data. We compared certificated values in the presence or absence of spectrum correction for NIST SRM 936a quinine sulfate. Comparing spectra in the literature or between other devices also demonstrates effectiveness of the correction.
