
Nobuaki Yamanaka
MD, PhD
Director: Tokyo Kidney Research Institute
Professor emeritus of Nippon Medical School
Histopathological diagnosis is now widely practiced in addition to general clinical tests for the diagnosis of renal disease. In the established methodology, the kidney tissue obtained from the needle biopsy are cut into three pieces, two of them are small and processed for examination by immunofluorescent microscopy and by transmission electron microscopy (TEM). The large remainder portion is embedded in paraffin and finely sliced, a stained specimen for light microscopy (LM) is prepared, and the histological changes are examined. The information obtained from these three methodologies is consolidated for the pathological diagnosis of the renal biopsy specimen.
TEM is widely used for kidney tissue examination, but the use of scanning electron microscopy (SEM) for examination of renal biopsy has remained largely unexplored. Reports on its use for biopsy specimen are small in number, and generally limited to research studies, due to the complexity of the sample preparation and the restricted information only on the images of tissue surfaces.
With the recent development of tabletop low-vacuum scanning electron microscope (LV-SEM), a new observation method has been investigated by utilizing their magnification and resolution levels, which are substantially higher than those of LM. This new technology in LV-SEM is direct observable means of ordinary glass-slide LM specimens with no further treatment, and it also provides the possibility of histopathological examination in 3D level according to the LM specimen thickness. Establishment of the examination method using LV-SEM holds promise of enhancing the quality of pathological diagnosis. Here we present some of the results of our investigation on examination of renal biopsy specimens by using LV-SEM, which may serve as reference for further progress.
Tabletop Microscope TM3030 developed by Hitachi High-Tech Corporation, with its small size, ease of operational freedom from a darkroom requirement, is highly practical. It performs the basic general functions of SEMs, and provides sample observation magnifications of up to 60,000×. It essentially consists of a small vacuum pump, an SEM unit small enough for tabletop placement, and a computer for operational control of the sample and observation image visualization, and it runs on an ordinary 100 V power supply (Fig. 1).
In noting the low-vacuum properties of this SEM, Dr. Sumire Inaga of the Department of Anatomy in the Faculty of Medicine at Tottori University established a transformational methodology by simply placing an ordinary glass-slide LM specimen in the LV-SEM specimen chamber. The tissue without a cover glass on the stained LM specimen can be examined, by irradiating it with an electron beam in the backscattered electron mode (BEM), and observing the resulting SEM image (Fig. 2).
The light microscopic examination of histopathological specimens generally involves staining the specimen with hematoxylin-eosin stain (HE stain). HE stain is being frequently used in conjunction with PAS stain, which effectively stains basement membrane, and Masson stain which is effective for assessment of fibrosis of the tissue. For renal biopsy examination, the standard practice has become to observe specimens stained with HE, PAS, and Masson (or AZAN) stains plus silver impregnation stain, periodic acid silver methenamine stain (PASM stain). PASM staining procedure exposes aldehyde groups by oxidizing polysaccharide-protein, which is the main constituent of basement membrane and other parts of the extracellular matrix. Reacting this with silver stains, the silver bonding region becomes dark brown, making the silver-negative cell components and silver-positive extracellular matrix components clearly distinguishable. HE stain is usually added as counterstaining to obtain the PASM-HE specimen. In this way, information is derived from both HE and silver staining. The method is highly effective for observing specimens with the light microscope1). PASM-HE stain has been widely used in pathological examination for kidney biopsy specimen.

Fig. 1 Low-vacuum SEM (LV-SEM) configuration

Fig. 2 Submission of LM specimen to LV-SEM
Clear contrast, which cannot be obtained in LV-SEM images with the usual stained specimens for LM observation, can be obtained by using a heavy-metal stain to image in the backscattered electron mode. This enables visualization and observation of fine details in the tissue structure, since the strength of the backscattered electron signal varies with the positive reaction of the heavy metal stain bonding to the tissue. As a silver impregnation stain, PASM can be used without modification in LV-SEM to obtain images clearly delineating basement membrane and other silver-positive components. In the renal disease, many cases are involved with glomerular lesion, and identification of lesions in the cell components and in the extracellular matrix, such as basement membrane and mesangial matrix, are often conclusive for diagnosis and pathological assessment. LV-SEM can be used to observe these fine lesions, and is therefore expected to contribute substantially to the quality of diagnosis. A remaining drawback, however, is poor contrast for the silver-negative structures in tissues, and the distinction of cell components and other primary tissue constituents is unclear. The assessment of cell components has therefore remained difficult.
Dr. Sumire Inaga of Tottori University has invented heavy-metal staining by platinum—what she terms “platinum-blue” staining—and has shown that it enables effective observation of the cell components2). Platinum-blue is characterized by cell-tissue specificity and effective strengthening of backscattered electron signals, which facilitate observation of paraffin-embedded tissue sections by LV-SEM. Staining with the platinum-blue solution after deparaffinization of an unstained thin paraffin section on a glass slide results in clear visualization of the cell structure of the LM specimen in LV-SEM observation. In this way, the platinum-blue-stained image provides clear visualization of the cell components and other constituents, while the PASM-stained image clearly visualizes the silver-positive basement membrane and other constituents. These staining characteristics are mutually complementary. With successive slicing of paraffin-embedded tissue for PASM-stained and platinum-blue-stained specimens and combination of LV-SEM images of both, the ultrafine structural image of tissue lesions in the same position can be closely examined (Fig. 3).
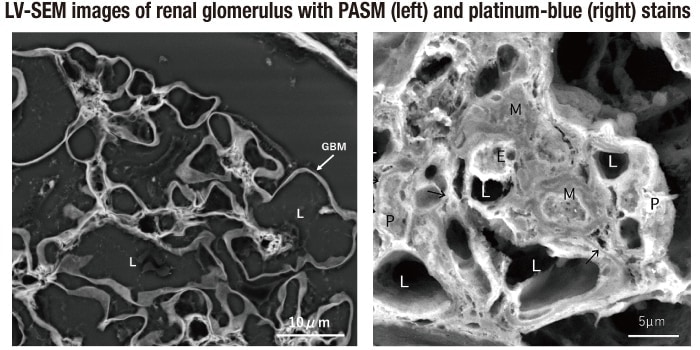
In PASM (left) stains, silver-positive components of the basement membrane and other constituents, complemented by the clear platinum-blue staining of the cell components (right; figure by Dr. Sumire Inaga).
GBM: basement membrane; L: vascular lumen; M: mesangial cell; E: endothelial cell; P: podocyte.
Fig. 3 PASM and platinum-blue stains
PASM-stained specimens, which are commonly used in renal biopsy diagnosis, can be directly observed by LV-SEM3). The difference between LM and LV-SEM images can be readily seen by comparing a low-power magnification LM image that shows an entire PASM-stained renal biopsy specimen and an LV-SEM image of the same specimen at the same magnification (Fig. 4). It is impossible to compare TEM images with those obtained by LM from the same biopsy specimens, but LV-SEM permits observation of exactly the same regions as those in the LM observation. This capability is highly significant, and opens the way to a higher level of precision in analysis of intratissue lesions. LV-SEM also permits observation of any region in the LM tissue specimen at any chosen magnification, thus further increasing its effectiveness. The highest magnification available with LM is about 600×, or 1,000× with an oil immersion lens. With LV-SEM, the same region can be magnified by up to 30,000× to 60,000×. Even more important is the outstanding precision provided by its 50 nm resolution, compared with the 200 nm resolution provided by LM. Its capability to provide 3D information on the entire specimen thickness further contributes to clear, detailed perception of the lesion.
These LV-SEM capabilities can often show clear abnormalities in observation of the same regions where LM for PASM-stained specimens cannot. Fig. 5, from a case exhibiting hematuria, shows the findings for a region in which the LM 600× magnification indistinctly shows the presence of an abnormality, whereas 3D observation at higher magnification by LV-SEM clearly confirms that it is a small rupture in the basement membrane that is leaking a blood cell.
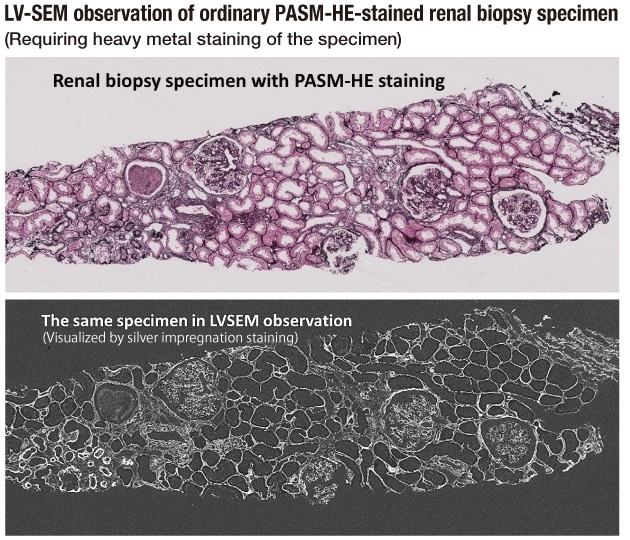
Fig. 4 Comparison of tissue images obtained by LM and LV-SEM from the same LM slide
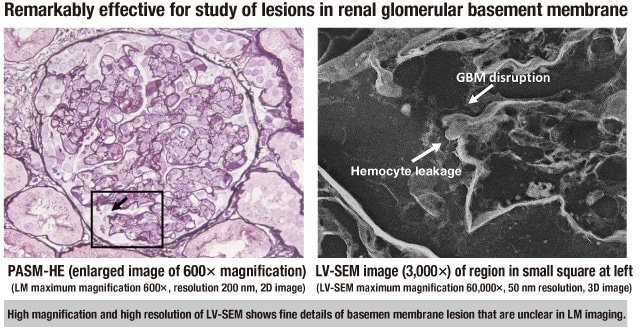
Fig. 5 Provision of high-precision information by LV-SEM
Thin basement membrane disease is a hereditary disease in which a hereditary abnormality in Type IV collagen composing the basement membrane induces thinning of the glomerular basement membrane, and is clinically characterized by persistent hematuria. LM findings can suggest some degree of basement membrane thinning, but conclusive diagnosis is established by immunostaining (usually immunofluorescence) of the Type IV collagen α chain subunits and measurement of the basement membrane thickness in the TEM image4). The presumed cause of hematuria is associated thinning of the basement membrane, but a conclusive finding of rupture or other disruption in the basement membrane has been elusive despite the large number of cases investigated by TEM, and the precise mechanism of hematuria has remained unclear. LV-SEM promises to provide new insights. When LV-SEM was applied to specimens that had been stained with PASM for LM examination in cases of thin basement membrane disease confirmed by genetic examination, immunostaining, and TEM, small cracks and pores throughout the thin glomerular basement membrane were observed, suggesting their strong association with hematuria (Fig. 6). TEM observations are inherently limited to small areas, but the capability of LV-SEM for examination throughout the expanse of the LM specimen together with 3D imaging through the entire specimen thickness enables this finding. At present, the number of cases investigated with LV-SEM has been small and further study will be necessary, but it holds strong promise for highly effective investigation and analysis of basement membrane lesions.
IgA nephropathy is a renal disease with a high incidence in Japan and Asia. It is characterized by deposition of IgA, an immunoglobulin present in the glomerulus, and is typically found in clinical cases of hematuria. The literature on research in many countries concerning its cause and the related pathology, therapeutic methods, and prognosis is voluminous, but key aspects of the mechanisms involved remain unclear. Reports on TEM investigation in many cases have suggested that thinning of the basement membrane is often observed and a few have confirmed a finding of tiny rupture in the basement membrane and images of erythrocyte leakage from the rupture, but their common association with hematuria remains to be elucidated4). We have therefore developed a new method of glomerular basement membrane investigation, in which PASM staining is performed over the fluorescence-stained specimens from confirmed cases of IgA nephropathy, which generally requires fluorescent evidence for diagnosis. These PASM-stained specimens were examined by LV-SEM5). The resulting LV-SEM 3D images clearly showed small crack and pore formations throughout the glomerular basement membrane in IgA nephropathy (Fig. 7), and clearly indicate that the formation of numerous small ruptures in fragile basement membrane is a cause of hematuria.
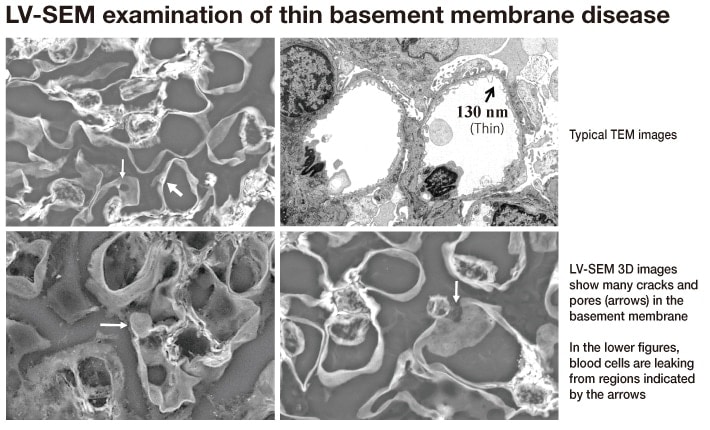
Fig. 6 LV-SEM images of glomeruli in thin basement membrane disease
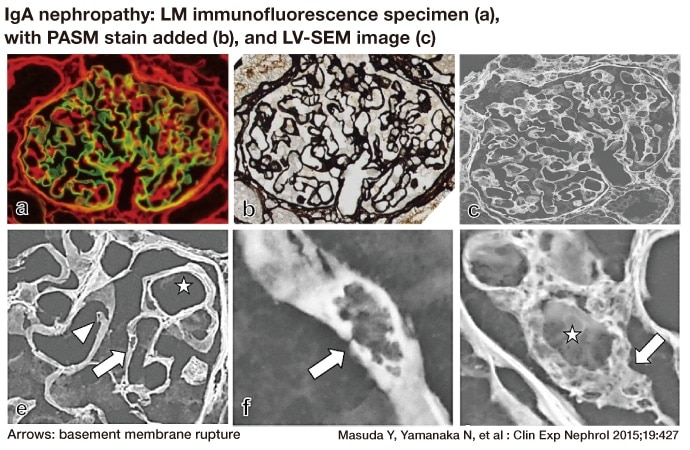
Fig. 7 Correspondence between LV-SEM images and LM immunofluorescence findings
Many cases of renal glomerular disease exhibit nephrotic syndrome with clinically prominent proteinuria, and the frequency of membranous nephropathy as the primary disease is high. In this disease, immune complex deposits are extensive along the renal glomerular basement membrane and result in structural changes in the basement membrane, accelerated protein leakage from the glomeruli, and induce marked proteinuria. For conclusive diagnosis, the general rule is confirmation of deposits consisting mainly of immunoglobulin IgG and complement component 3 (C3) on the glomerular basement membrane by immunofluorescence. TEM images show deposits on the sub-epithelial surfaces of the basement membrane that are embedded in what appear to be protrusions (“spikes”) formed by basement membrane components. In LM specimen examination of membranous nephropathy, what appear to be numerous spinous protrusions called spikes, which stain dark brown, are found in the PASM stain specimen on the outer surface of the basement membrane4). In observation by LV-SEM, on the other hand, the PASM-stained LM specimens in the advanced stage of disease show indistinct or transparent complexes deposited on the silver negative epithelial cells, and clear details of changes in the silver positive basement membrane. In 3D images obtained by LV-SEM, moreover, unlike the images that appear in 2D TEM images as basement membrane spikes, it can be seen that the basement membrane components have actually extended and encircled the deposit in reaction to deposited immunocomplexes, thus forming craters (Fig. 8). The 3D examination of renal biopsy LM specimens by LV-SEM thus shows details of changes in tissue that were previously unrecognized or misunderstood, and thus may lead to key revisions in the concept of renal disease lesions.
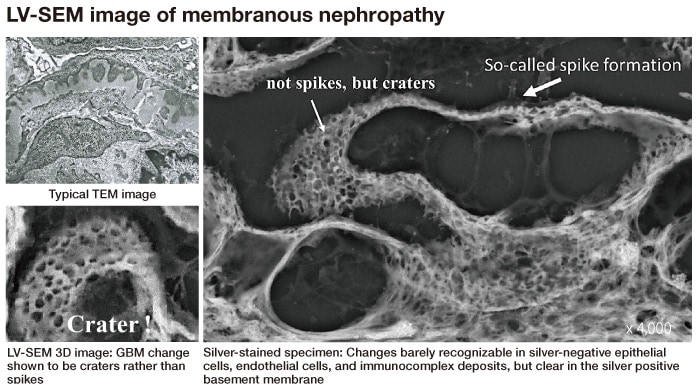
Fig. 8 Modification of the existing concept by LV-SEM examination
In renal biopsy diagnosis, interstitial tissue fibrosis often poses particular problems. The appropriate therapeutic approach and the prognosis are greatly affected by its level, since the decline in renal function progresses rapidly when fibrosis is prominent, and investigation of its cause and assessment of the pathological state are therefore crucial. Assessment of fibrosis is usually based on the color tones and extent of Masson or Azan staining, but differences in color tone and other aspects vary with specimen quality, which thus affects the assessment. Interstitial fibrosis results from an increase in collagen fiber, which is positive also for PASM staining. In LV-SEM observation of the PASM-stained specimen, the collagen fiber becomes increasingly conspicuous with the progression of fibrosis, and thus enables close, detailed assessment of the fibrosis pathology (Fig. 9, left). It has also been shown in LV-SEM observation that in some cases where the changes were identified as fibrosis by existing Masson and other staining methods, the cause was not an increase in collagen fiber but rather an increase in non-fibrotic matrix components, thus indicating a need to reconsider the assessment of fibrosis by LM observation (Fig. 9, right).
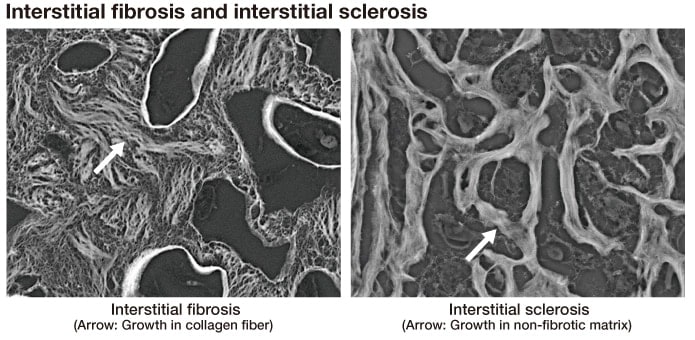
Fig. 9 Distinguishing interstitial sclerosis from interstitial fibrosis
Interstitial infiltration by inflammatory cells sometimes becomes prominent in conjunction with interstitial lesions. Precise identification of the cell type is occasionally difficult, but is facilitated by platinum-blue staining in LV-SEM observation. The cells can also be distinguished to a considerable degree by LV-SEM observation of PASM-stained specimens, which exhibit slight contrasts in their blood cell components (Fig. 10). PASM staining is commonly performed on LM specimens for renal biopsy examination, and they can be directly observed by LV-SEM after simply removing the cover glass. LV-SEM is thus useful and effective in cases requiring identification of infiltrating inflammatory cells.
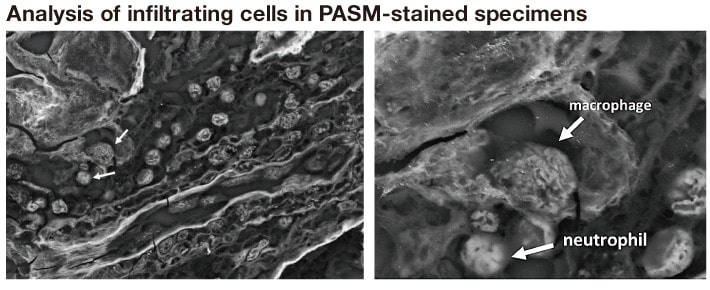
Fig. 10 Identification of infiltrating blood cells
In developing the practical application of LV-SEM examination of LM specimens for pathological diagnosis, it is essential to compare the 2D information on lesions obtained by TEM and the corresponding 3D information obtained by LV-SEM for the same positions.
In the comparison shown here, the PASM-stained LM specimen was first observed by LV-SEM. It was then embedded in epoxy resin (Epon), and after polymerization, was sliced into ultrathin sections as specimens for TEM observation of the same position as that observed by LV-SEM (Fig. 11). Comparisons of the information by such a way will be essential for advancement of the analytical examination.
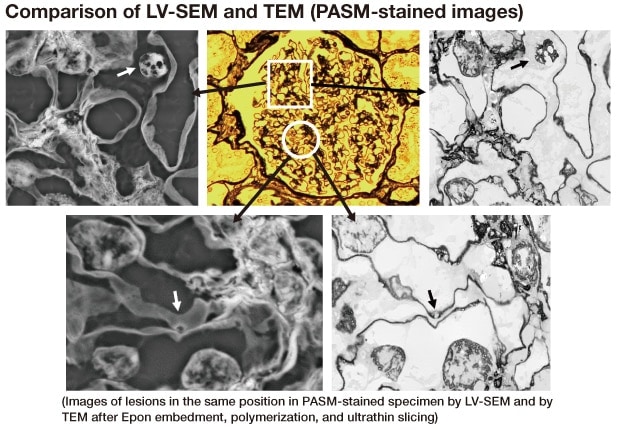
Fig. 11 Comparison of LV-SEM and TEM images
LV-SEM can be effectively used to observe LM specimens of renal biopsy and any other tissue with appropriate stain selection. Its application may well be extended to the situation where tissue examination by usual transmission electron microscopes is unavailable, in case of tumor difficult to diagnose in usual LM examination, and many other circumstances which need ultramicroscopic information. With the expanding adoption of LV-SEM, new staining techniques in addition to platinum-blue staining and PASM silver staining will also emerge and development of applications will be extended to cytopathological diagnosis, rapid analysis to meet urgent needs, and other requirements. It will serve as a major contribution to improving accuracy in pathological tissue diagnosis.
Practical development of diagnosis by LV-SEM examination with LM specimens will require extensive comparative studies of LV-SEM images for LM and TEM images for the same specimens. The construction of a large database for consolidating their findings and the results of examinations for various types of lesions should be established. The LV-SEM methodology has originated in Japan, but this construction and its widespread adoption will require many cooperative working in concert across many borders.
References
This database is available in our membership website "S.I.navi" at the following URL.
See more