3. Colorimetric Analysis (2)
Neglecting cleaning increases absorbance (Bouger-Beer's law).
Are you a tropical fish-lover? Caring for an aquarium is somewhat troublesome, although the fish are lovely.
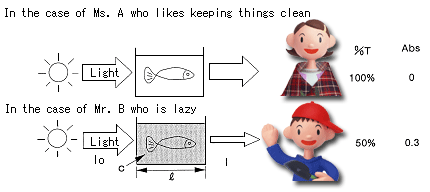
Here are Ms. A and Mr. B who keep tropical fish. Ms. A likes keeping things clean, and the aquarium in which the tropical fish are kept is always clear and beautiful. On the other hand, Mr. B is lazy, and the aquarium tends to become muddy.
The aquariums of these two persons are placed by the window. Since the water of Ms. A's aquarium is clear, the light that comes in from a window can mostly be seen even through the aquarium. However, since the water of Mr. B's aquarium becomes muddy, light hardly comes in through the aquarium.
Of the light that comes in from the window, the percent of light that comes in through the aquarium is called "transmittance (T)."
Generally, transmittance is more commonly expressed as a percentage "transmittance (%T)". The part of the light that comes in from the window and is absorbed into the water of the aquarium is calls "absorbance (Abs)."
The water of the aquarium that is not cleaned becomes muddy and absorbs light preventing its transmittance, resulting in an increase in absorbance and a decrease in transmittance. In contrast, since the water of the aquarium that is always clean clear lets the light pass through well, the transmittance is close to 100%, and the absorbance is close to zero.
Let's make this theory into a mathematical formula. When the light that comes in from the window is defined Io, and the light that comes out from the aquarium is defined as I, the transmittance is I/Io. Furthermore, when the width of the aquarium is defined as 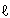 (optical path length), the degree of muddiness of the water (concentration) as c, and a constant (called "molar absorption coefficient") unique to the substance (in this case, the substance that causes muddiness) as ε, the transmittance will become 10-
(optical path length), the degree of muddiness of the water (concentration) as c, and a constant (called "molar absorption coefficient") unique to the substance (in this case, the substance that causes muddiness) as ε, the transmittance will become 10-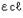 (Formula 1).
(Formula 1).
Absorbance can be obtained with the formula 3, since it is a logarithm of the inverse of the transmittance, suggesting that the absorbance is proportional to the molar absorption coefficient, concentration, and optical path length. This formula 3 is called Bouger-Beer's law.
Bouger-Beer's law
The absorbance (Abs) of a solute (coloring substance) is proportional to the concentration of a solution (c) and the thickness of a solution layer (l).
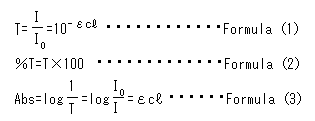
Notes 1) Bouger-Beer's law had been called "Lambert-Beer's law".
Notes 2) Molar absorption coefficient is a constant unique to a substance (solute), and is needed to quantify each substance by spectrometry.
