6. Structure of a spectrophotometer (1)
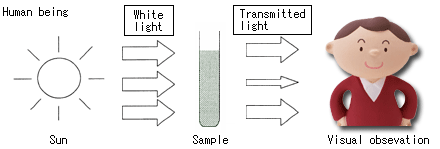
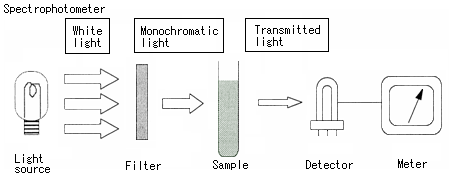
Differences between a man and a spectrophotometer (rough structure)
How can we know whether a color is deep or light, which color (wavelength) this substance absorbs in the wavelength range (200 - 800 nm) of the ultraviolet-visible range that we have studied until now?
Even human beings can roughly estimate the absorption wavelength from the color of a substance, or recognize color density, etc., however, we cannot know the exact wavelength, and also we have individual differences. Besides, the ultraviolet range is not visible to our eyes.
Here, an "ultraviolet-visible spectrophotometer," a device that can measure even in the ultraviolet range without individual differences, comes in.
With the spectrophotometer, we measure a color (wavelength) using an artificial source instead of the sun, and a detector and a meter instead of our eyes. The biggest difference between the spectrophotometer and macroscopic observation is that the spectrophotometer measures the degree of absorption of each color, using prism or diffraction grating (not irradiating white light on a substance directly) to split the light into each color and irradiate each of the colors (monochromatic light) on the substance in order (called "scanning").
The spectrophotometer splits white light into monochromatic lights to measure absorption.