3. Principle of the Atomic Absorption Photometer
Q: "What does an atomic absorption photometer use to analyze?"
A: Like a spectrophotometer, an atomic absorption photometer measures how much light is absorbed when the light flux from a light source passes through a material to be measured. The fundamental difference from a spectrophotometer is the condition of the material to be measured. That is, an atomic absorption photometer analyzes using atomic absorption, while a spectrophotometer analyzes using molecular photoabsorption.
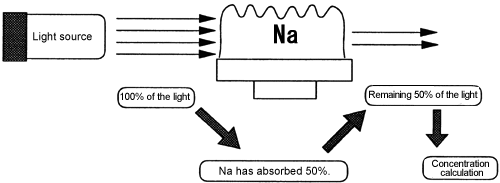
Q: "What is atomic absorption?"
A:It means that an atom absorbs (eats) a particular wavelength. For example, Na eats only light at the wavelength of 589.0 nm.

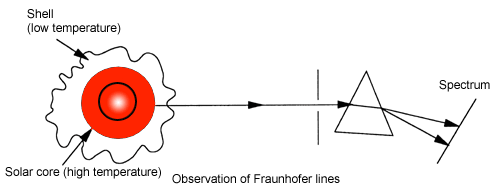
As for discovery of atomic absorption, A long time ago…in the early 19th century, a man named Fraunhofer observed the spectrum of sunlight and found dark lines in it. These lines were named Fraunhofer lines after the discoverer of the dark lines. In the middle of the 19th century, Kirchhoff inferred that Fraunhofer lines might be due to absorption by atoms.
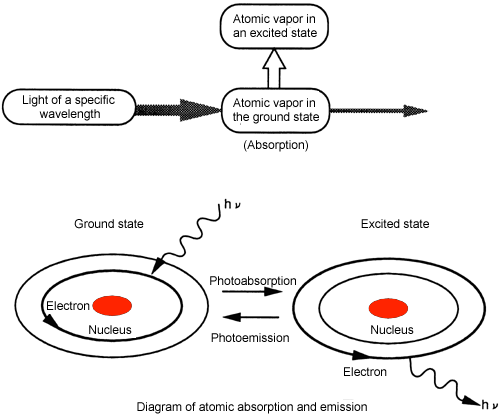
Q: "Why does atomic absorption take place?"
A: An atom usually exists in its lowest, stable energy level (the ground state). However, ... An atomic vapor in the ground state will be converted into an atomic vapor in an excited state by irradiation of light at a particular wavelength. A part of the irradiated light will be consumed. This is called atomic absorption.
Simple diagram showing atomic energy levels
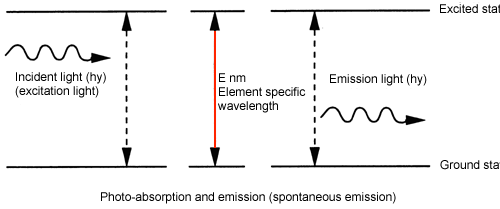
Do you now understand atomic absorption?
To induce atomic absorption in a system and measure its amount ...
