4. About a light source and a specimen chamber
1. Light source ...
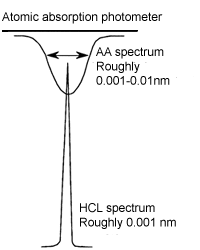
The spectral width of an atomic absorption spectrum is very narrow (usually about 0.01 nm). To measure atomic absorption, a light source with narrower spectral width is necessary. The spectral width of the light source of the spectrophotometer is as wide as 1-2 nm.
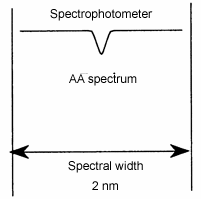
Therefore, an atomic absorption cannot be measured with a continuum source. In an atomic absorption photometer, a hollow cathode lamp (HCL) is used. The spectral width of an emission line (bright line) of a hollow cathode lamp is even narrower than a line in an atomic absorption spectrum.

2. Specimen chamber...
A specimen has to be changed to an atomic state to perform atomic absorption. However, metallic elements do not exist in the atomic state in the specimen. In order convert the specimen into the atomic state, an outside force is necessary. So, the specimen is burned, changing it into the atomic state, using a burner, etc. (absorption cannot be measured simply by injecting the specimen into a cell, as with a spectrophotometer, because the specimen is not atomized only by injection.)
Atomization by an external force such as heat
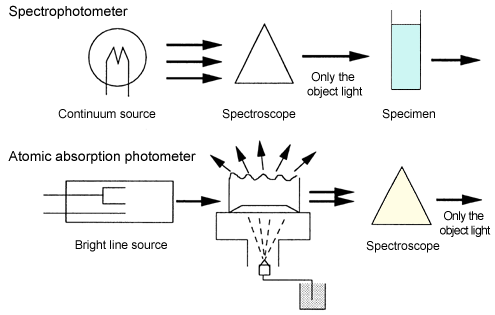
Here is the answer to why the spectroscope is located behind the specimen chamber (atomization section). In a spectrophotometer, a spectroscope is located in front of a specimen chamber, to let only the object light of the continuous light transmit. On the other hand, in an atomic absorption photometer, the spectroscope is located behind the specimen chamber to remove the light component (luminescence component) of the flame generated in the atomization section.
The method using a burner (flame method) is not the only means of atomizing (atomization method). The various types will be explained, later.
