Analysis of Bis(2-ethylhexyl) phthalate(DEHP) in Drinking Water
Recently, an overseas news article reported that alarge amount of Bis(2-ethylhexyl) phthalate (DEHP), which is mainly used as a plasticizer, had been detected in food, which brought about a level of higher consciousness regarding food safety. This plasticizer is normally added to plastics to make them soft or easy to fabricate; however, if taken into the human body in largeamounts, it is considered to constitute a cancer-causing hazard and to present a high risk of liver function disorders. It is also specified as a target of the Restriction of Hazardous Substances (RoHS) Directive by the European Union (EU).
Commercial sports drinks usually include an emulsifier to avoid turbidity, which is also used in jellies, health foods,etc. The most widely used emulsifiers are gum arabic and palm oil. However, in this study it was found that manufacturers had been using DEHP, which is banned for use in food, in place of palm oil for the purpose of cost reduction. Moreover, as a result ofa follow-up investigation, DEHP use was also found in beverages, fruit juice, jams, etc.which were distributed in the market, attracting substantial attention from the media. Accordingly, it is now important to confirm whether or not DEHP is included in a wide variety of foods and materials.
Methods for detecting phthalate esters include the LC Method, LC-MSMethod,etc.
Here, we will introduce the LC-DAD Method.
Analysis of Bis (2-ethylhexyl) phthalate (DEHP)
Analysis of standard samples
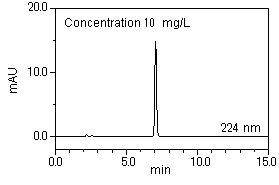
Sample preparation: Adjust the concentration to 0.1 - 100 mg/L using acetonitrile.
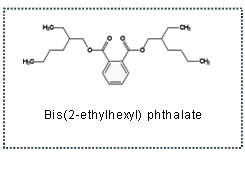
[Standard sample] Bis (2-ethylhexyl) phthalate
[Chemical structural formula]
[Contour indication and extraction chromatogram]
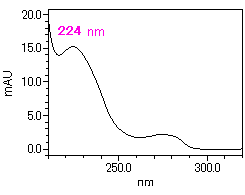
[Absorption spectrum of DEHP]
Conditions
| Column | HITACHI LaChrom C18 (5 µm) (4.6 mmI.D. x 150 mm) |
|---|---|
| Mobile Phase | H2O/CH3CN = 2/98 (v/v) |
| Flow Rate | 1.0 mL/min |
| Column Temperature | 30°C |
| Detection | DAD 224 nm |
| Injection vol. | 10 µL |
System configuration
5110 Pump
5210 AutoSampler
5310 Column Oven
5430 Diode Array Detector
Empower2 Data Processing System
Calibration curve and determination limit
[Calibration curve]
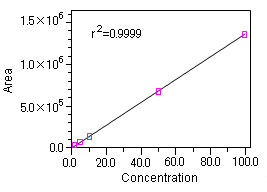
The calibration curve shows good linearity in the concentration range from 0.1 to 100 mg/L.
Note that the determination limit of this measurement method is 0.18 mg/L, when S/N = 10.
Repeatability of standard sample
Repeatability of the standard sample (concentration: 10 mg/L) over 6 repeated measurements is shown below.
| Retention time (min) | Area | |
|---|---|---|
| 1 | 7.058 | 131486 |
| 2 | 7.057 | 131742 |
| 3 | 7.056 | 131456 |
| 4 | 7.057 | 131769 |
| 5 | 7.056 | 131609 |
| 6 | 7.057 | 131743 |
| Mean | 7.057 | 131634 |
| %RSD | 0.011 | 0.11 |
Good repeatability results were obtained for both retention time (0.011%) and peak area (0.11%).
An example of sample analysis
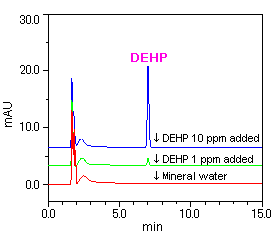
[Sample(1): Mineral water] [Sample(2): Sports drink]
DEHP (prepared using acetonitrile) was added to samples (1) and (2) to produce the analysis samples.
(Pre-treatment: Filtered with a 0.45µm filter)
[Sample(1): Mineral water]
[Sample(2): Sports drink]
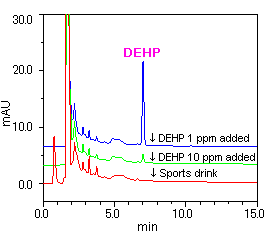
Samples (1) and (2) were first analyzed without added DEHP, and no DEHP was detected in either sample. Afterwards, samples (1) and (2) were again analyzed with the addition of 1 ppm and 10 ppm of DEHP, and the detection of DEHP was confirmed in each case.
NOTE:
These data are an example of measurement; the individual values cannot be guaranteed.
The system is for research use only, and is not intended for any animal or human therapeutic or diagnostic use.
In order to read a PDF file, you need to have Adobe® Reader®
